Targeted expression of a multifunctional chimeric neurotrophin in the lesioned sciatic nerve accelerates regeneration of sensory and motor axons
- PMID: 9560265
- PMCID: PMC20250
- DOI: 10.1073/pnas.95.9.5269
Targeted expression of a multifunctional chimeric neurotrophin in the lesioned sciatic nerve accelerates regeneration of sensory and motor axons
Abstract
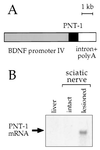
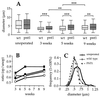
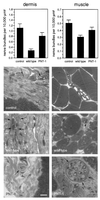
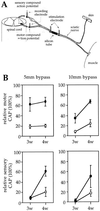
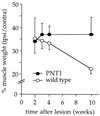
Peripheral nerve injury markedly regulates expression of neurotrophins and their receptors in the lesioned nerve. However, the role of endogenously produced neurotrophins in the process of nerve regeneration is unclear. Expression of a multifunctional neurotrophin, pan-neurotrophin-1 (PNT-1), was targeted to the peripheral nerves of transgenic mice by using a gene promoter that is specifically activated after nerve lesion but that is otherwise silent in all other tissues and during development. PNT-1 is a chimeric neurotrophin that combines the active sites of the neurotrophins nerve growth factor, brain-derived neurotrophic factor, and neurotrophin-3 and binds and activates all known neurotrophin receptors. In adult transgenic mice, PNT-1 was highly expressed in transected but not in intact sciatic nerve. Morphometric analyses at the electron microscopy level showed increased and accelerated recovery of axon diameter of myelinated fibers in crushed peripheral nerves of transgenic mice compared with wild type. Examination of nerve bundles in target tissues indicated accelerated reinnervation of foot pad dermis and flexor plantaris muscle in transgenic mice. Moreover, transected sensory and motor axons of transgenic mice showed faster and increased return of neurophysiological responses, suggesting an accelerated rate of axonal elongation. Importantly, transgenic mice also showed a markedly ameliorated loss of skeletal muscle weight, indicating functional regeneration of motor axons. Together, these data provide evidence, at both the anatomical and functional levels, that neurotrophins endogenously produced by the lesioned nerve are capable of significantly accelerating the regeneration of both sensory and motor axons after peripheral nerve damage. In addition, our results indicate that exogenous PNT-1 administration may be an effective therapeutic treatment of peripheral nerve injuries.
Figures
Similar articles
-
Faster nerve regeneration after sciatic nerve injury in mice over-expressing basic fibroblast growth factor.J Neurobiol. 2006 Aug;66(9):940-8. doi: 10.1002/neu.20265. J Neurobiol. 2006. PMID: 16758491
-
Preferential Enhancement of Sensory and Motor Axon Regeneration by Combining Extracellular Matrix Components with Neurotrophic Factors.Int J Mol Sci. 2016 Dec 29;18(1):65. doi: 10.3390/ijms18010065. Int J Mol Sci. 2016. PMID: 28036084 Free PMC article.
-
Long-term effects of deprivation of cell support in the distal stump on peripheral nerve regeneration.J Neurosci Res. 1994 Sep 1;39(1):23-30. doi: 10.1002/jnr.490390104. J Neurosci Res. 1994. PMID: 7807589
-
Specificity of peripheral nerve regeneration: interactions at the axon level.Prog Neurobiol. 2012 Jul;98(1):16-37. doi: 10.1016/j.pneurobio.2012.05.005. Epub 2012 May 15. Prog Neurobiol. 2012. PMID: 22609046 Review.
-
Putative roles of soluble trophic factors in facial nerve regeneration, target reinnervation, and recovery of vibrissal whisking.Exp Neurol. 2018 Feb;300:100-110. doi: 10.1016/j.expneurol.2017.10.029. Epub 2017 Nov 8. Exp Neurol. 2018. PMID: 29104116 Review.
Cited by
-
Analysis of gene expression following sciatic nerve crush and spinal cord hemisection in the mouse by microarray expression profiling.Cell Mol Neurobiol. 2001 Oct;21(5):497-508. doi: 10.1023/a:1013867306555. Cell Mol Neurobiol. 2001. PMID: 11860187 Free PMC article.
-
Neurotrophic factors and their receptors in axonal regeneration and functional recovery after peripheral nerve injury.Mol Neurobiol. 2003 Jun;27(3):277-324. doi: 10.1385/MN:27:3:277. Mol Neurobiol. 2003. PMID: 12845152 Review.
-
Neural and molecular features on Charcot-Marie-Tooth disease plasticity and therapy.Neural Plast. 2012;2012:171636. doi: 10.1155/2012/171636. Epub 2012 Jun 13. Neural Plast. 2012. PMID: 22745917 Free PMC article. Review.
-
Tryptophan 2,3-dioxygenase is a key modulator of physiological neurogenesis and anxiety-related behavior in mice.Mol Brain. 2009 Mar 27;2:8. doi: 10.1186/1756-6606-2-8. Mol Brain. 2009. PMID: 19323847 Free PMC article.
-
Tissue plasminogen activator-mediated fibrinolysis protects against axonal degeneration and demyelination after sciatic nerve injury.J Cell Biol. 2000 May 29;149(5):1157-66. doi: 10.1083/jcb.149.5.1157. J Cell Biol. 2000. PMID: 10831618 Free PMC article.
References
-
- Ibáñez C F, Hökfelt T, Olson L, Fuxe K, Jörnvall H, Ottoson D. Wenner-Gren Center International Series. Vol. 67. Oxford: Elsevier; 1995. , 472 pp.
-
- Barde Y-A. Neuron. 1989;2:1525–1534. - PubMed
-
- Thoenen H. Science. 1995;270:593–598. - PubMed
-
- Lewin G, Barde Y-A. Annu Rev Neurosci. 1996;19:289–317. - PubMed
-
- Crowley C, Spencer S D, Nishimura M C, Chen K S, Pittsmeek S, Armanini M P, Ling L H, Mcmahon S B, Shelton D L, Levinson A D, Phillips H S. Cell. 1994;76:1001–1011. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

