Direct visualization of antigen-specific CD8+ T cells during the primary immune response to Epstein-Barr virus In vivo
- PMID: 9565632
- PMCID: PMC2212279
- DOI: 10.1084/jem.187.9.1395
Direct visualization of antigen-specific CD8+ T cells during the primary immune response to Epstein-Barr virus In vivo
Abstract
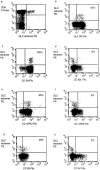
Primary infection with virus can stimulate a vigorous cytotoxic T cell response. The magnitude of the antigen-specific component versus the bystander component of a primary T cell response remains controversial. In this study, we have used tetrameric major histocompatibility complex-peptide complexes to directly visualize antigen-specific cluster of differentration (CD)8+ T cells during the primary immune response to Epstein-Barr virus (EBV) infection in humans. We show that massive expansion of activated, antigen-specific T cells occurs during the primary response to this virus. In one individual, T cells specific for a single EBV epitope comprised 44% of the total CD8+ T cells within peripheral blood. The majority of the antigen-specific cells had an activated/memory phenotype, with expression of human histocompatibility leukocyte antigen (HLA) DR, CD38, and CD45RO, downregulation of CD62 leukocyte (CD62L), and low levels of expression of CD45RA. After recovery from AIM, the frequency of antigen-specific T cells fell in most donors studied, although populations of antigen-specific cells continued to be easily detectable for at least 3 yr.
Figures


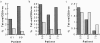

References
-
- Deckhut AM, Allan W, McMickle A, Eihelberger M, Blackman MA, Doherty PC, Woodland DL. Prominent usage of V beta 8.3 T cells in the H-2D-b restricted response to an influenza A virus nucleoprotein epitope. J Immunol. 1993;151:2658–2666. - PubMed
-
- McHeyzer-Williams MG, Davis MM. Antigen specific development of primary and memory T cells in vivo. Science. 1995;268:106–111. - PubMed
-
- Callan MFC, Steven N, Krausa P, Wilson JDK, Moss PAH, Gillespie GM, Bell JI, McMichael AJ. Large clonal expansions of CD8+T cells in acute infectious mononucleosis. Nat Med. 1996;2:906–911. - PubMed
-
- Pantaleo B, Demarest JF, Soudeyns H, Groziosi C, Denis F, Adelsberger JW, Borros P, Saag MS, Shaw GM, Sekaly RP, Fauci AS. Major expansions of CD8+ T cells with a predominant Vβ usage during the primary immune response to HIV. Nature. 1994;370:463–467. - PubMed
-
- Maryanski J, Jongeneel CV, Bucher P, Casanova JL, Walker PR. Single cell PCR analysis of TCR repertoires selected by antigen in vivo: a high magnitude CD8 response is comprised of very few clones. Immunity. 1996;4:47–54. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

