Role of dimerization of the membrane-associated growth factor kit ligand in juxtacrine signaling: the Sl17H mutation affects dimerization and stability-phenotypes in hematopoiesis
- PMID: 9565637
- PMCID: PMC2212272
- DOI: 10.1084/jem.187.9.1451
Role of dimerization of the membrane-associated growth factor kit ligand in juxtacrine signaling: the Sl17H mutation affects dimerization and stability-phenotypes in hematopoiesis
Abstract
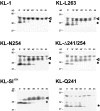
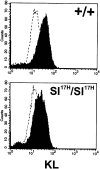
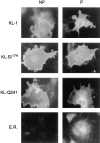
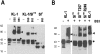
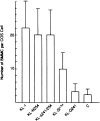
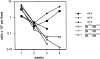
The Kit ligand (KL)/Kit receptor pair functions in hematopoiesis, gametogenesis, and melanogenesis. KL is encoded at the murine steel (Sl) locus and encodes a membrane growth factor which may be proteolytically processed to produce soluble KL. The membrane-associated form of KL is critical in mediating Kit function in vivo. Evidence for a role of cytoplasmic domain sequences of KL comes from the Sl17H mutation, a splice site mutation that replaces the cytoplasmic domain with extraneous amino acids. Using deletion mutants and the Sl17H allele, we have investigated the role of the cytoplasmic domain sequences of KL in biosynthetic processing and cell surface presentation. The normal KL protein products are processed for cell surface expression, where they form dimers. Both Sl17H and the cytoplasmic deletion mutants of KL were processed to the cell surface; however, the rate of transport and protein stability were affected by the mutations. Deletion of cytoplasmic domain sequences of KL did not affect dimerization of KL. In contrast, dimerization of the Sl17H protein was reduced substantially. In addition, we have characterized the hematopoietic cell compartment in Sl17H mutant mice. The Sl17H mutation has only minor effects on hematopoiesis. Tissue and peritoneal mast cell numbers were reduced in mutant mice as well as in myeloid progenitors. Interestingly, long-term bone marrow cultures from Sl17H mice did not sustain the long-term production of hematopoietic cells. In addition, homing of normal hematopoietic progenitors to the spleen of irradiated Sl17H/Sl17H recipient mice was diminished in transplantation experiments, providing evidence for a role of Kit in homing or lodging. These results demonstrate that the membrane forms of KL exist as homodimers on the cell surface and that dimerization may play an important role in KL/Kit-mediated juxtacrine signaling.
Figures



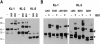
References
-
- Lee DC, Fenton SE, Berkowitz EA, Hissong MA. Transforming growth factor: expression, regulation, and biological activities. Pharmacol Rev. 1995;47:51–85. - PubMed
-
- Bosenberg MW, Massague J. Juxtacrine cell signaling molecules. Curr Opin Cell Biol. 1993;5:823–838. - PubMed
-
- Hill RJ, Sternberg PW. The gene lin-3 encodes an inductive signal for vulval development in C. elegans. . Nature. 1992;358:470–476. - PubMed
-
- Hart AC, Kramer H, Van Vacter DL, Jr, Paidhungat M, Zipursky SL. Induction of cell fate in the Drosophilaretina: the bride of sevenless protein is predicted to contain a large extracellular domain and seven transmembrane segments. Genes Dev. 1990;4:1835–1847. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

