Rho1p-Bni1p-Spa2p interactions: implication in localization of Bni1p at the bud site and regulation of the actin cytoskeleton in Saccharomyces cerevisiae
- PMID: 9571251
- PMCID: PMC25343
- DOI: 10.1091/mbc.9.5.1221
Rho1p-Bni1p-Spa2p interactions: implication in localization of Bni1p at the bud site and regulation of the actin cytoskeleton in Saccharomyces cerevisiae
Abstract
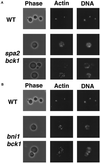
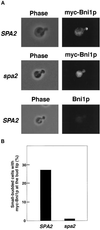
Rho1p is a yeast homolog of mammalian RhoA small GTP-binding protein. Rho1p is localized at the growth sites and required for bud formation. We have recently shown that Bni1p is a potential target of Rho1p and that Bni1p regulates reorganization of the actin cytoskeleton through interactions with profilin, an actin monomer-binding protein. Using the yeast two-hybrid screening system, we cloned a gene encoding a protein that interacted with Bni1p. This protein, Spa2p, was known to be localized at the bud tip and to be implicated in the establishment of cell polarity. The C-terminal 254 amino acid region of Spa2p, Spa2p(1213-1466), directly bound to a 162-amino acid region of Bni1p, Bni1p(826-987). Genetic analyses revealed that both the bni1 and spa2 mutations showed synthetic lethal interactions with mutations in the genes encoding components of the Pkc1p-mitogen-activated protein kinase pathway, in which Pkc1p is another target of Rho1p. Immunofluorescence microscopic analysis showed that Bni1p was localized at the bud tip in wild-type cells. However, in the spa2 mutant, Bni1p was not localized at the bud tip and instead localized diffusely in the cytoplasm. A mutant Bni1p, which lacked the Rho1p-binding region, also failed to be localized at the bud tip. These results indicate that both Rho1p and Spa2p are involved in the localization of Bni1p at the growth sites where Rho1p regulates reorganization of the actin cytoskeleton through Bni1p.
Figures



Similar articles
-
Dynamic localization and function of Bni1p at the sites of directed growth in Saccharomyces cerevisiae.Mol Cell Biol. 2001 Feb;21(3):827-39. doi: 10.1128/MCB.21.3.827-839.2001. Mol Cell Biol. 2001. PMID: 11154270 Free PMC article.
-
Bni1p implicated in cytoskeletal control is a putative target of Rho1p small GTP binding protein in Saccharomyces cerevisiae.EMBO J. 1996 Nov 15;15(22):6060-8. EMBO J. 1996. PMID: 8947028 Free PMC article.
-
Bni1p and Bnr1p: downstream targets of the Rho family small G-proteins which interact with profilin and regulate actin cytoskeleton in Saccharomyces cerevisiae.EMBO J. 1997 May 15;16(10):2745-55. doi: 10.1093/emboj/16.10.2745. EMBO J. 1997. PMID: 9184220 Free PMC article.
-
Ras-related GTPases and the cytoskeleton.Mol Biol Cell. 1992 May;3(5):475-9. doi: 10.1091/mbc.3.5.475. Mol Biol Cell. 1992. PMID: 1611153 Free PMC article. Review.
-
The protein kinase C-mediated MAP kinase pathway involved in the maintenance of cellular integrity in Saccharomyces cerevisiae.Mol Microbiol. 1999 May;32(4):671-80. doi: 10.1046/j.1365-2958.1999.01375.x. Mol Microbiol. 1999. PMID: 10361272 Review.
Cited by
-
Cdc42p regulation of the yeast formin Bni1p mediated by the effector Gic2p.Mol Biol Cell. 2012 Oct;23(19):3814-26. doi: 10.1091/mbc.E12-05-0400. Epub 2012 Aug 23. Mol Biol Cell. 2012. PMID: 22918946 Free PMC article.
-
A comparative sequence analysis reveals a common GBD/FH3-FH1-FH2-DAD architecture in formins from Dictyostelium, fungi and metazoa.BMC Genomics. 2005 Mar 1;6:28. doi: 10.1186/1471-2164-6-28. BMC Genomics. 2005. PMID: 15740615 Free PMC article.
-
The yeast actin cytoskeleton: from cellular function to biochemical mechanism.Microbiol Mol Biol Rev. 2006 Sep;70(3):605-45. doi: 10.1128/MMBR.00013-06. Microbiol Mol Biol Rev. 2006. PMID: 16959963 Free PMC article. Review.
-
The Glc7p-interacting protein Bud14p attenuates polarized growth, pheromone response, and filamentous growth in Saccharomyces cerevisiae.Eukaryot Cell. 2002 Dec;1(6):884-94. doi: 10.1128/EC.1.6.884-894.2002. Eukaryot Cell. 2002. PMID: 12477789 Free PMC article.
-
The novel adaptor protein, Mti1p, and Vrp1p, a homolog of Wiskott-Aldrich syndrome protein-interacting protein (WIP), may antagonistically regulate type I myosins in Saccharomyces cerevisiae.Genetics. 2002 Mar;160(3):923-34. doi: 10.1093/genetics/160.3.923. Genetics. 2002. PMID: 11901111 Free PMC article.
References
-
- Castrillon DH, Wasserman SA. Diaphanous is required for cytokinesis in Drosophila and shares domains of similarity with the products of the limb deformity gene. Development. 1994;120:3367–3377. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

