The Us9 gene product of pseudorabies virus, an alphaherpesvirus, is a phosphorylated, tail-anchored type II membrane protein
- PMID: 9573219
- PMCID: PMC109967
- DOI: 10.1128/JVI.72.6.4560-4570.1998
The Us9 gene product of pseudorabies virus, an alphaherpesvirus, is a phosphorylated, tail-anchored type II membrane protein
Abstract
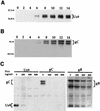
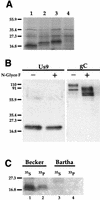
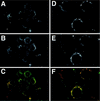
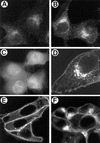
The Us9 gene is highly conserved among the alphaherpesviruses sequenced to date, yet its function remains unknown. In this report, we demonstrate that the pseudorabies virus (PRV) Us9 protein is present in infected cell lysates as several phosphorylated polypeptides ranging from 17 to 20 kDa. Synthesis is first detected at 6 h postinfection and is sensitive to the DNA synthesis inhibitor phosphonoacetic acid. Unlike the herpes simplex virus type 1 Us9 homolog, which was reported to be associated with nucleocapsids in the nuclei of infected cells (M. C. Frame, D. J. McGeoch, F. J. Rixon, A. C. Orr, and H. S. Marsden, Virology 150:321-332, 1986), PRV Us9 localizes to the secretory pathway (predominately to the Golgi apparatus) and not to the nucleus. By fusing the enhanced green fluorescent protein (EGFP) reporter molecule to the carboxy terminus of Us9, we demonstrated that Us9 not only is capable of targeting a Us9-EGFP fusion protein to the Golgi compartment but also is able to direct efficient incorporation of such chimeric molecules into infectious viral particles. Moreover, through protease digestion experiments with Us9-EGFP-containing viral particles, we demonstrated that the Us9 protein is inserted into the viral envelope as a type II, tail-anchored membrane protein.
Figures




References
-
- Babic N, Klupp B, Brack A, Mettenleiter T C, Ugolini G, Flamand A. Deletion of glycoprotein gE reduces the propagation of pseudorabies virus in the nervous system of mice after intranasal inoculation. Virology. 1996;219:279–284. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

