Overexpression of nitrate reductase in tobacco delays drought-induced decreases in nitrate reductase activity and mRNA
- PMID: 9576799
- PMCID: PMC35015
- DOI: 10.1104/pp.117.1.293
Overexpression of nitrate reductase in tobacco delays drought-induced decreases in nitrate reductase activity and mRNA
Abstract
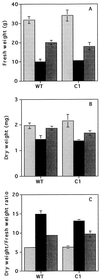
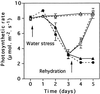
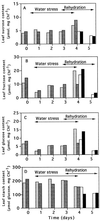
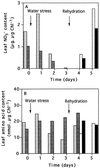
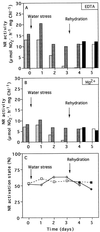
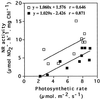
Transformed (cauliflower mosaic virus 35S promoter [35S]) tobacco (Nicotiana plumbaginifolia L.) plants constitutively expressing nitrate reductase (NR) and untransformed controls were subjected to drought for 5 d. Drought-induced changes in biomass accumulation and photosynthesis were comparable in both lines of plants. After 4 d of water deprivation, a large increase in the ratio of shoot dry weight to fresh weight was observed, together with a decrease in the rate of photosynthetic CO2 assimilation. Foliar sucrose increased in both lines during water stress, but hexoses increased only in leaves from untransformed controls. Foliar NO3- decreased rapidly in both lines and was halved within 2 d of the onset of water deprivation. Total foliar amino acids decreased in leaves of both lines following water deprivation. After 4 d of water deprivation no NR activity could be detected in leaves of untransformed plants, whereas about 50% of the original activity remained in the leaves of the 35S-NR transformants. NR mRNA was much more stable than NR activity. NR mRNA abundance increased in the leaves of the 35S-NR plants and remained constant in controls for the first 3 d of drought. On the 4th d, however, NR mRNA suddenly decreased in both lines. Rehydration at d 3 caused rapid recovery (within 24 h) of 35S-NR transcripts, but no recovery was observed in the controls. The phosphorylation state of the protein was unchanged by long-term drought. There was a strong correlation between maximal extractable NR activity and ambient photosynthesis in both lines. We conclude that drought first causes increased NR protein turnover and then accelerates NR mRNA turnover. Constitutive NR expression temporarily delayed drought-induced losses in NR activity. 35S-NR expression may therefore allow more rapid recovery of N assimilation following short-term water deficit.
Figures

Similar articles
-
Drought-induced effects on nitrate reductase activity and mRNA and on the coordination of nitrogen and carbon metabolism in maize leaves.Plant Physiol. 1998 May;117(1):283-92. doi: 10.1104/pp.117.1.283. Plant Physiol. 1998. PMID: 9576798 Free PMC article.
-
Growth of tobacco in short-day conditions leads to high starch, low sugars, altered diurnal changes in the Nia transcript and low nitrate reductase activity, and inhibition of amino acid synthesis.Planta. 1998 Dec;207(1):27-41. doi: 10.1007/s004250050452. Planta. 1998. PMID: 9951717
-
Tobacco mutants with a decreased number of functional nia genes compensate by modifying the diurnal regulation of transcription, post-translational modification and turnover of nitrate reductase.Planta. 1997;203(3):304-19. doi: 10.1007/s004250050196. Planta. 1997. PMID: 9431679
-
Negative regulation of nitrate reductase gene expression by glutamine or asparagine accumulating in leaves of sulfur-deprived tobacco.Planta. 2000 Sep;211(4):587-95. doi: 10.1007/s004250000322. Planta. 2000. PMID: 11030559
-
Mechanism and importance of post-translational regulation of nitrate reductase.J Exp Bot. 2004 Jun;55(401):1275-82. doi: 10.1093/jxb/erh132. Epub 2004 Apr 23. J Exp Bot. 2004. PMID: 15107452 Review.
Cited by
-
Nitrogen uptake, assimilation and remobilization in plants: challenges for sustainable and productive agriculture.Ann Bot. 2010 Jun;105(7):1141-57. doi: 10.1093/aob/mcq028. Epub 2010 Mar 18. Ann Bot. 2010. PMID: 20299346 Free PMC article. Review.
-
Carbon/Nitrogen Imbalance Associated with Drought-Induced Leaf Senescence in Sorghum bicolor.PLoS One. 2015 Aug 28;10(8):e0137026. doi: 10.1371/journal.pone.0137026. eCollection 2015. PLoS One. 2015. PMID: 26317421 Free PMC article.
-
Genetic and physiological analysis of germination efficiency in maize in relation to nitrogen metabolism reveals the importance of cytosolic glutamine synthetase.Plant Physiol. 2002 Dec;130(4):1860-70. doi: 10.1104/pp.009647. Plant Physiol. 2002. PMID: 12481069 Free PMC article.
-
Improving coordination of plant growth and nitrogen metabolism for sustainable agriculture.aBIOTECH. 2020 Aug 31;1(4):255-275. doi: 10.1007/s42994-020-00027-w. eCollection 2020 Oct. aBIOTECH. 2020. PMID: 36304130 Free PMC article. Review.
-
Two cytosolic glutamine synthetase isoforms of maize are specifically involved in the control of grain production.Plant Cell. 2006 Nov;18(11):3252-74. doi: 10.1105/tpc.106.042689. Epub 2006 Nov 30. Plant Cell. 2006. PMID: 17138698 Free PMC article.
References
-
- Becker TW, Fock HP. The activity of nitrate reductase and some pool sizes of some amino acids and some sugars in water-stressed maize leaves. Photosynth Res. 1986a;8:267–274. - PubMed
-
- Becker TW, Fock HP. Effects of water stress on the gas exchange, the activities of some enzymes of carbon and nitrogen metabolism, and on the pool sizes of some organic acids in maize leaves. Photosynth Res. 1986b;8:175–181. - PubMed
-
- Beyrouty CA, Grigg BC, Norman RJ, Wells BR. Nutrient uptake by rice in response to water management. J Plant Nutr. 1994;8:39–55.
LinkOut - more resources
Full Text Sources
Other Literature Sources

