Phosphorylation and association with the transcription factor Atf1 regulate localization of Spc1/Sty1 stress-activated kinase in fission yeast
- PMID: 9585506
- PMCID: PMC316836
- DOI: 10.1101/gad.12.10.1464
Phosphorylation and association with the transcription factor Atf1 regulate localization of Spc1/Sty1 stress-activated kinase in fission yeast
Abstract
Control of gene expression by stress-activated protein kinase (SAPK) cascades is crucial for combating cytotoxic stress. Elements of these cascades have been investigated in detail, but regulation of stress signal transduction from the cytoplasm to the nucleus is poorly understood. Herein are reported subcellular localization studies of fission yeast Spc1, a homolog of human p38 and budding yeast Hog1p SAPKs. Stress induces transient nuclear localization of Spc1. Nuclear translocation of Spc1 is coupled with disassociation from its activator kinase Wis1. However, Spc1 does not concentrate in the nucleus of Deltawis1 cells; therefore Wis1 does not tether Spc1 in the cytoplasm. Unphosphorylatable forms of Spc1 are dispersed in the cytoplasm and nucleus, even in cells that also produce wild-type Spc1. Thus, Spc1 must be phosphorylated by Wis1 to localize in the nucleus. Nuclear retention of Spc1 requires Atf1, a transcription factor that is the key nuclear substrate of Spc1. Nuclear localization of Atf1 requires Pcr1, a heterodimerization partner of Atf1. These studies show that phosphorylation and association with Atf1 are required for nuclear localization of Spc1.
Figures


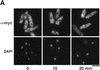

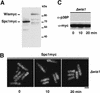








Comment in
-
SAPKs and transcription factors do the nucleocytoplasmic tango.Genes Dev. 1998 May 15;12(10):1391-7. doi: 10.1101/gad.12.10.1391. Genes Dev. 1998. PMID: 9585499 Review. No abstract available.
References
-
- Alfa C, Fantes P, Hyams J, McLeod M, Warbrick E. Experiments with fission yeast. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1993.
-
- Basi G, Schmid E, Maundrell K. TATA box mutations in the Schizosaccharomyces pombe nmt1 promoter affect transcription efficiency but not the transcription start point or thiamine repressibility. Gene. 1993;123:131–136. - PubMed
-
- Canagarajah BJ, Khokhlatchev A, Cobb MH, Goldsmith EJ. Activation mechanism of the MAP kinase ERK2 by dual phosphorylation. Cell. 1997;90:859–869. - PubMed
-
- Cano E, Mahadevan LC. Parallel signal processing among mammalian MAPKs. Trends Biochem Sci. 1995;20:117–122. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
