Two amino acid substitutions convert a guanylyl cyclase, RetGC-1, into an adenylyl cyclase
- PMID: 9600905
- PMCID: PMC27573
- DOI: 10.1073/pnas.95.11.5993
Two amino acid substitutions convert a guanylyl cyclase, RetGC-1, into an adenylyl cyclase
Abstract
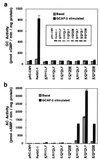
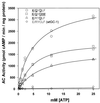
Guanylyl cyclases (GCs) and adenylyl cyclases (ACs) have fundamental roles in a wide range of cellular processes. Whereas GCs use GTP as a substrate to form cGMP, ACs catalyze the analogous conversion of ATP to cAMP. Previously, a model based on the structure of adenylate cyclase was used to predict the structure of the nucleotide-binding pocket of a membrane guanylyl cyclase, RetGC-1. Based on this model, we replaced specific amino acids in the guanine-binding pocket of GC with their counterparts from AC. A change of two amino acids, E925K together with C995D, is sufficient to completely alter the nucleotide specificity from GTP to ATP. These experiments strongly validate the AC-derived RetGC-1 structural model and functionally confirm the role of these residues in nucleotide discrimination.
Figures

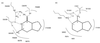
Similar articles
-
Substrate specificity determinants of class III nucleotidyl cyclases.FEBS J. 2016 Oct;283(20):3723-3738. doi: 10.1111/febs.13837. Epub 2016 Sep 9. FEBS J. 2016. PMID: 27542992
-
Catalytic mechanism of the adenylyl and guanylyl cyclases: modeling and mutational analysis.Proc Natl Acad Sci U S A. 1997 Dec 9;94(25):13414-9. doi: 10.1073/pnas.94.25.13414. Proc Natl Acad Sci U S A. 1997. PMID: 9391039 Free PMC article.
-
Exchange of substrate and inhibitor specificities between adenylyl and guanylyl cyclases.J Biol Chem. 1998 Jun 26;273(26):16332-8. doi: 10.1074/jbc.273.26.16332. J Biol Chem. 1998. PMID: 9632695
-
Regulation of photoreceptor membrane guanylyl cyclases by guanylyl cyclase activator proteins.Methods. 1999 Dec;19(4):521-31. doi: 10.1006/meth.1999.0894. Methods. 1999. PMID: 10581151 Review.
-
Sequence homologies between guanylyl cyclases and structural analogies to other signal-transducing proteins.FEBS Lett. 1991 Mar 25;280(2):301-6. doi: 10.1016/0014-5793(91)80317-v. FEBS Lett. 1991. PMID: 1672855 Review.
Cited by
-
Establishment and characterization of a new class of adenylate cyclases (class VII ACs) in plants.Heliyon. 2023 Aug 2;9(8):e18612. doi: 10.1016/j.heliyon.2023.e18612. eCollection 2023 Aug. Heliyon. 2023. PMID: 37593644 Free PMC article.
-
The crystal structure of the catalytic domain of a eukaryotic guanylate cyclase.BMC Struct Biol. 2008 Oct 7;8:42. doi: 10.1186/1472-6807-8-42. BMC Struct Biol. 2008. PMID: 18842118 Free PMC article.
-
Structural basis for inhibition of mammalian adenylyl cyclase by calcium.Biochemistry. 2009 Apr 21;48(15):3387-97. doi: 10.1021/bi802122k. Biochemistry. 2009. PMID: 19243146 Free PMC article.
-
Determining consequences of retinal membrane guanylyl cyclase (RetGC1) deficiency in human Leber congenital amaurosis en route to therapy: residual cone-photoreceptor vision correlates with biochemical properties of the mutants.Hum Mol Genet. 2013 Jan 1;22(1):168-83. doi: 10.1093/hmg/dds421. Epub 2012 Oct 3. Hum Mol Genet. 2013. PMID: 23035049 Free PMC article.
-
Evaluating the role of retinal membrane guanylyl cyclase 1 (RetGC1) domains in binding guanylyl cyclase-activating proteins (GCAPs).J Biol Chem. 2015 Mar 13;290(11):6913-24. doi: 10.1074/jbc.M114.629642. Epub 2015 Jan 23. J Biol Chem. 2015. PMID: 25616661 Free PMC article.
References
-
- Eckstein F, Romaniuk P J, Heideman W, Storm D R. J Biol Chem. 1981;256:9118–9120. - PubMed
-
- Gerlt J A, Coderre J A, Wolin M S. J Biol Chem. 1980;255:331–334. - PubMed
-
- Senter P D, Eckstein F. J Biol Chem. 1983;258:6741–6745. - PubMed
-
- Koch K-W, Eckstein F, Stryer L. J Biol Chem. 1990;265:9659–9663. - PubMed
-
- Beuve A, Danchin A. J Mol Biol. 1992;225:933–938. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

