Stepwise in vitro affinity maturation of Vitaxin, an alphav beta3-specific humanized mAb
- PMID: 9600913
- PMCID: PMC27581
- DOI: 10.1073/pnas.95.11.6037
Stepwise in vitro affinity maturation of Vitaxin, an alphav beta3-specific humanized mAb
Abstract
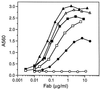
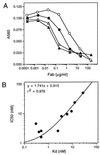
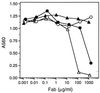
A protein engineering strategy based on efficient and focused mutagenesis implemented by codon-based mutagenesis was developed. Vitaxin, a humanized version of the antiangiogenic antibody LM609 directed against a conformational epitope of the alphav beta3 integrin complex, was used as a model system. Specifically, focused mutagenesis was used in a stepwise fashion to rapidly improve the affinity of the antigen binding fragment by greater than 90-fold. In the complete absence of structural information about the Vitaxin-alphav beta3 interaction, phage-expressed antibody libraries for all six Ig heavy and light chain complementarity-determining regions were expressed and screened by a quantitative assay to identify variants with improved binding to alphav beta3. The Vitaxin variants in these libraries each contained a single mutation, and all 20 amino acids were introduced at each complementarity-determining region residue, resulting in the expression of 2,336 unique clones. Multiple clones displaying 2- to 13-fold improved affinity were identified. Subsequent expression and screening of a library of 256 combinatorial variants of the optimal mutations identified from the primary libraries resulted in the identification of multiple clones displaying greater than 50-fold enhanced affinity. These variants inhibited ligand binding to receptor more potently as demonstrated by inhibition of cell adhesion and ligand competition assays. Because of the limited mutagenesis and combinatorial approach, Vitaxin variants with enhanced affinity were identified rapidly and required the synthesis of only 2,592 unique variants. The use of such small focused libraries obviates the need for phage affinity selection approaches typically used, permitting the use of functional assays and the engineering of proteins expressed in mammalian cell culture.
Figures
Similar articles
-
Humanization of a murine monoclonal antibody by simultaneous optimization of framework and CDR residues.J Mol Biol. 1999 Nov 19;294(1):151-62. doi: 10.1006/jmbi.1999.3141. J Mol Biol. 1999. PMID: 10556035
-
Affinity maturation of a humanized rat antibody for anti-RAGE therapy: comprehensive mutagenesis reveals a high level of mutational plasticity both inside and outside the complementarity-determining regions.J Mol Biol. 2009 May 8;388(3):541-58. doi: 10.1016/j.jmb.2009.03.019. Epub 2009 Mar 13. J Mol Biol. 2009. PMID: 19285987
-
Enhancement of scFv fragment reactivity with target antigens in binding assays following mixing with anti-tag monoclonal antibodies.J Immunol Methods. 2004 Nov;294(1-2):23-35. doi: 10.1016/j.jim.2004.08.005. J Immunol Methods. 2004. PMID: 15604013
-
Cognizance of Molecular Methods for the Generation of Mutagenic Phage Display Antibody Libraries for Affinity Maturation.Int J Mol Sci. 2019 Apr 15;20(8):1861. doi: 10.3390/ijms20081861. Int J Mol Sci. 2019. PMID: 30991723 Free PMC article. Review.
-
In vitro affinity maturation of HuCAL antibodies: complementarity determining region exchange and RapMAT technology.Immunotherapy. 2009 Jul;1(4):571-83. doi: 10.2217/imt.09.23. Immunotherapy. 2009. PMID: 20635988 Review.
Cited by
-
In vivo near-infrared fluorescence imaging of integrin alphavbeta3 in an orthotopic glioblastoma model.Mol Imaging Biol. 2006 Nov-Dec;8(6):315-23. doi: 10.1007/s11307-006-0059-y. Mol Imaging Biol. 2006. PMID: 17053862
-
ITGB3/CD61: a hub modulator and target in the tumor microenvironment.Am J Transl Res. 2019 Dec 15;11(12):7195-7208. eCollection 2019. Am J Transl Res. 2019. PMID: 31934272 Free PMC article. Review.
-
Effective Optimization of Antibody Affinity by Phage Display Integrated with High-Throughput DNA Synthesis and Sequencing Technologies.PLoS One. 2015 Jun 5;10(6):e0129125. doi: 10.1371/journal.pone.0129125. eCollection 2015. PLoS One. 2015. PMID: 26046845 Free PMC article.
-
A general method for greatly improving the affinity of antibodies by using combinatorial libraries.Proc Natl Acad Sci U S A. 2005 Jun 14;102(24):8466-71. doi: 10.1073/pnas.0503543102. Epub 2005 Jun 6. Proc Natl Acad Sci U S A. 2005. PMID: 15939870 Free PMC article.
-
Differential regulation of monocyte cytokine release by αV and β(2) integrins that bind CD23.Immunology. 2012 Jun;136(2):241-51. doi: 10.1111/j.1365-2567.2012.03576.x. Immunology. 2012. PMID: 22348662 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

