The serine proteinase inhibitor (serpin) plasminogen activation inhibitor type 2 protects against viral cytopathic effects by constitutive interferon alpha/beta priming
- PMID: 9607921
- PMCID: PMC2212304
- DOI: 10.1084/jem.187.11.1799
The serine proteinase inhibitor (serpin) plasminogen activation inhibitor type 2 protects against viral cytopathic effects by constitutive interferon alpha/beta priming
Abstract
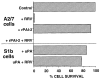
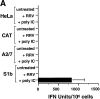
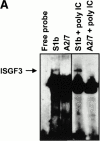
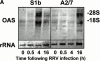
The serine proteinase inhibitor (serpin) plasminogen activator inhibitor type 2 (PAI-2) is well characterized as an inhibitor of extracellular urokinase-type plasminogen activator. Here we show that intracellular, but not extracellular, PAI-2 protected cells from the rapid cytopathic effects of alphavirus infection. This protection did not appear to be related to an effect on apoptosis but was associated with a PAI-2-mediated induction of constitutive low-level interferon (IFN)-alpha/beta production and IFN-stimulated gene factor 3 (ISGF3) activation, which primed the cells for rapid induction of antiviral genes. This primed phenotype was associated with a rapid development of resistance to infection by the PAI-2 transfected cells and the establishment of a persistent productive infection. PAI-2 was also induced in macrophages in response to viral RNA suggesting that PAI-2 is a virus response gene. These observations, together with the recently demonstrated PAI-2-mediated inhibition of tumor necrosis factor-alpha induced apoptosis, (a) illustrate that PAI-2 has an additional and distinct function as an intracellular regulator of signal transduction pathway(s) and (b) demonstrate a novel activity for a eukaryotic serpin.
Figures









References
-
- Remold-O'Donnell E. The ovalbumin family of serpin proteins. FEBS Lett. 1993;315:105–108. - PubMed
-
- Ray CA, Black RA, Kronheim SR, Greenstreet TA, Sleath PR, Salvesen GS, Pickup DJ. Viral inhibition of inflammation: cowpox virus encodes an inhibitor of the interleukin-1β converting enzyme. Cell. 1992;69:597–604. - PubMed
-
- Zou Z, Anisowicz A, Hendrix MJC, Thor A, Neveu M, Sheng S, Rafidi K, Seftor E, Sager R. Maspin, a serpin with tumor-suppressing activity in human mammary epithelial cells. Science. 1994;263:526–529. - PubMed
-
- Scott FL, Coughlin PB, Bird C, Cerruti L, Hayman JA, Bird P. Protease inhibitor 6 cannot be secreted, which suggests it's a new type of cellular serpin. J Biol Chem. 1996;271:1605–1612. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

