Cloning, expression, and characterization of the squid Na+-Ca2+ exchanger (NCX-SQ1)
- PMID: 9607941
- PMCID: PMC2217150
- DOI: 10.1085/jgp.111.6.857
Cloning, expression, and characterization of the squid Na+-Ca2+ exchanger (NCX-SQ1)
Abstract
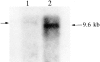
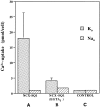
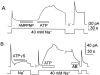
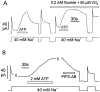
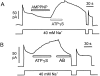
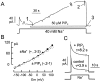
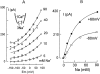
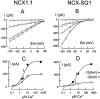
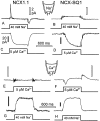
We have cloned the squid neuronal Na+-Ca2+ exchanger, NCX-SQ1, expressed it in Xenopus oocytes, and characterized its regulatory and ion transport properties in giant excised membrane patches. The squid exchanger shows 58% identity with the canine Na+-Ca2+ exchanger (NCX1.1). Regions determined to be of functional importance in NCX1 are well conserved. Unique among exchanger sequences to date, NCX-SQ1 has a potential protein kinase C phosphorylation site (threonine 184) between transmembrane segments 3 and 4 and a tyrosine kinase site in the Ca2+ binding region (tyrosine 462). There is a deletion of 47 amino acids in the large intracellular loop of NCX-SQ1 in comparison with NCX1. Similar to NCX1, expression of NCX-SQ1 in Xenopus oocytes induced cytoplasmic Na+-dependent 45Ca2+ uptake; the uptake was inhibited by injection of Ca2+ chelators. In giant excised membrane patches, the NCX-SQ1 outward exchange current showed Na+-dependent inactivation, secondary activation by cytoplasmic Ca2+, and activation by chymotrypsin. The NCX-SQ1 exchange current was strongly stimulated by both ATP and the ATP-thioester, ATP gamma S, in the presence of F- (0.2 mM) and vanadate (50 microM), and both effects reversed on application of a phosphatidylinositol-4',5'-bisphosphate antibody. NCX1 current was stimulated by ATP, but not by ATP gamma S. Like NCX1 current, NCX-SQ1 current was strongly stimulated by phosphatidylinositol-4',5'-bisphosphate liposomes. In contrast to results in squid axon, NCX-SQ1 was not stimulated by phosphoarginine (5-10 mM). After chymotrypsin treatment, both the outward and inward NCX-SQ1 exchange currents were more strongly voltage dependent than NCX1 currents. Ion concentration jump experiments were performed to estimate the relative electrogenicity of Na+ and Ca2+ transport reactions. Outward current transients associated with Na+ extrusion were much smaller for NCX-SQ1 than NCX1, and inward current transients associated with Ca2+ extrusion were much larger. For NCX-SQ1, charge movements of Ca2+ transport could be defined in voltage jump experiments with a low cytoplasmic Ca2+ (2 microM) in the presence of high extracellular Ca2+ (4 mM). The rates of charge movements showed "U"-shaped dependence on voltage, and the slopes of both charge-voltage and rate-voltage relations (1,600 s-1 at 0 mV) indicated an apparent valency of -0.6 charges for the underlying reaction. Evidently, more negative charge moves into the membrane field in NCX-SQ1 than in NCX1 when ions are occluded into binding sites.
Figures
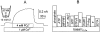
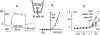




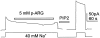
References
-
- Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

