Lung lymphocytes proliferate minimally in the murine pulmonary immune response to intratracheal sheep erythrocytes
- PMID: 9618385
- PMCID: PMC4123639
- DOI: 10.1165/ajrcmb.18.6.3063
Lung lymphocytes proliferate minimally in the murine pulmonary immune response to intratracheal sheep erythrocytes
Abstract
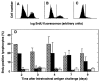
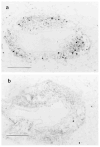
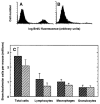
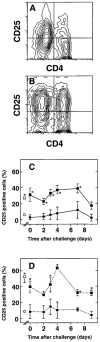
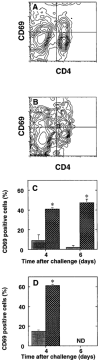
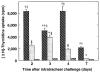
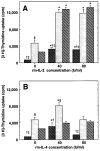
The importance of in situ lymphocyte proliferation for net accumulation of lung lymphocytes during pulmonary immune responses and in immunologic lung diseases remains uncertain. Accordingly, we studied the experimental pulmonary immune response of antigen-primed C57BL/6 mice to intratracheal challenge with the particulate antigen sheep red blood cells. Uptake of nucleotide analogs (bromodeoxyuridine in vivo and tritiated thymidine in vitro), expression of the cell activation antigens CD25 and CD69 by flow cytometry, and response to the antimitotic agent hydroxyurea (in vivo) were measured. Although many lung lymphocytes and CD4+ T cells were CD25+ and CD69+, indicating recent activation, all techniques demonstrated that lung lymphocytes proliferated minimally in vivo. Blockade of cell division by hydroxyurea administration for 24 h did not significantly decrease lung lymphocyte accumulation on Day 3 after challenge. Lung lymphocytes also proliferated minimally in vitro (even on macrophage removal and despite addition of exogenous interleukin [IL]-2 or IL-4). However, lung lymphocytes responded vigorously to mitogens (immobilized anti-CD3, phytohemagglutinin, or concanavalin A), excluding global unresponsiveness to restimulation. Thus, in this model of pulmonary immunity, accumulation of lung lymphocytes does not require local T-cell proliferation and presumably depends instead on recruitment.
Figures


Similar articles
-
Adhesion receptor phenotypes of murine lung CD4+ T cells during the pulmonary immune response to sheep erythrocytes.Am J Respir Cell Mol Biol. 1995 May;12(5):520-30. doi: 10.1165/ajrcmb.12.5.7537969. Am J Respir Cell Mol Biol. 1995. PMID: 7537969
-
Lung lymphocyte elimination by apoptosis in the murine response to intratracheal particulate antigen.J Clin Invest. 1997 Mar 1;99(5):1082-91. doi: 10.1172/JCI119236. J Clin Invest. 1997. PMID: 9062367 Free PMC article.
-
Lymphocyte activation in the lungs of SP-D null mice.Am J Respir Cell Mol Biol. 2002 Jul;27(1):24-33. doi: 10.1165/ajrcmb.27.1.4563. Am J Respir Cell Mol Biol. 2002. PMID: 12091242
-
Differential effect of CD69 targeting on bystander and antigen-specific T cell proliferation.J Leukoc Biol. 2012 Jul;92(1):145-58. doi: 10.1189/jlb.1011499. Epub 2012 Apr 27. J Leukoc Biol. 2012. PMID: 22544938
-
Lymphocyte-endothelial cell adhesive interactions in lung immunity: lessons from the murine response to particulate antigen.Immunopharmacology. 2000 Jul 25;48(3):223-9. doi: 10.1016/s0162-3109(00)00221-6. Immunopharmacology. 2000. PMID: 10960661 Review.
Cited by
-
Deficient in vitro and in vivo phagocytosis of apoptotic T cells by resident murine alveolar macrophages.J Immunol. 2000 Aug 15;165(4):2124-33. doi: 10.4049/jimmunol.165.4.2124. J Immunol. 2000. PMID: 10925298 Free PMC article.
-
Subset-specific reductions in lung lymphocyte accumulation following intratracheal antigen challenge in endothelial selectin-deficient mice.J Immunol. 2002 Sep 1;169(5):2570-9. doi: 10.4049/jimmunol.169.5.2570. J Immunol. 2002. PMID: 12193727 Free PMC article.
-
Effects of Anti-IL-17 on Inflammation, Remodeling, and Oxidative Stress in an Experimental Model of Asthma Exacerbated by LPS.Front Immunol. 2018 Jan 5;8:1835. doi: 10.3389/fimmu.2017.01835. eCollection 2017. Front Immunol. 2018. PMID: 29379497 Free PMC article.
-
Review: Collectins link innate and adaptive immunity in allergic airway disease.Innate Immun. 2010 Jun;16(3):183-90. doi: 10.1177/1753425910368446. Epub 2010 Apr 23. Innate Immun. 2010. PMID: 20418258 Free PMC article. Review.
-
Alcohol exacerbates murine pulmonary tuberculosis.Infect Immun. 2004 May;72(5):2556-63. doi: 10.1128/IAI.72.5.2556-2563.2004. Infect Immun. 2004. PMID: 15102763 Free PMC article.
References
-
- Berman JS, Beer DJ, Theodore AC, Kornfeld H, Bernardo J, Center DM. Lymphocyte recruitment to the lung. Am Rev Respir Dis. 1990;142:238–257. - PubMed
-
- Sitrin RG, Ansfield MJ, Kaltreider HB. The effect of pulmonary surface-active material on the generation and expression of murine B- and T- lymphocyte effector functions in vitro. Exp Lung Res. 1985;9:85–97. - PubMed
-
- Roth MD, Golub SH. Human pulmonary macrophages utilize prostaglandins and transforming growth factor β1 to suppress lymphocyte activation. J Leukocyte Biol. 1993;53:366–371. - PubMed
-
- Paine R, III, Chavis A, Gaposchkin D, Christensen P, Mody CH, Turka LA, Toews GB. A factor secreted by a human pulmonary alveolar epithelial-like cell line blocks T-cell proliferation between G1 and S phase. Am J Respir Cell Mol Biol. 1992;6:658–666. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

