Reexamination of the mechanism of hydroxyl radical adducts formed from the reaction between familial amyotrophic lateral sclerosis-associated Cu,Zn superoxide dismutase mutants and H2O2
- PMID: 9618471
- PMCID: PMC22595
- DOI: 10.1073/pnas.95.12.6675
Reexamination of the mechanism of hydroxyl radical adducts formed from the reaction between familial amyotrophic lateral sclerosis-associated Cu,Zn superoxide dismutase mutants and H2O2
Abstract
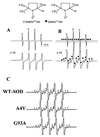
Amyotrophic lateral sclerosis (ALS) involves the progressive degeneration of motor neurons in the spinal cord and motor cortex. Mutations to Cu,Zn superoxide dismutase (SOD) linked with familial ALS are reported to increase hydroxyl radical adduct formation from hydrogen peroxide as measured by spin trapping with 5, 5'-dimethyl-1-pyrrolline N-oxide (DMPO). In the present study, we have used oxygen-17-enriched water and H2O2 to reinvestigate the mechanism of DMPO/.OH formation from the SOD and SOD mutants. The relative ratios of DMPO/.17OH and DMPO/.16OH formed in the Fenton reaction were 90% and 10%, respectively, reflecting the ratios of H217O2 to H216O2. The reaction of the WT SOD with H217O2 in bicarbonate/CO2 buffer yielded 63% DMPO/.17OH and 37% DMPO/.16OH. Similar results were obtained from the reaction between familial ALS SOD mutants and H217O2: DMPO/.17OH (64%); DMPO/.16OH (36%) from A4V and DMPO/.17OH (62%); and DMPO/.16OH (38%) from G93A. These results were confirmed further by using 5-diethoxyphosphoryl-5-methyl-1-pyrroline N-oxide spin trap, a phosphorylated analog of DMPO. Contrary to earlier reports, the present results indicate that a significant fraction of DMPO/.OH formed during the reaction of SOD and familial ALS SOD mutants with H2O2 is derived from the incorporation of oxygen from water due to oxidation of DMPO to DMPO/.OH presumably via DMPO radical cation. No differences were detected between WT and mutant SODs, neither in the concentration of DMPO/.OH or DEPMPO/.OH formed nor in the relative incorporation of oxygen from H2O2 or water.
Figures


References
-
- Price D L, Cleveland D W, Koliatsos V E. Neurobiol Dis. 1994;1:3–11. - PubMed
-
- Rosen D R, Siddique T, Patterson D, Figlewicz D A, Sapp P, Hentati A, Donaldson D, Goto J, O’Regan J P, Deng H X, et al. Nature (London) 1993;362:59–62. - PubMed
-
- Bruijn L I, Cleveland D W. Neuropath Appl Neurobiol. 1996;22:373–387. - PubMed
-
- Cudkowicz M E, McKenna-Yasek D, Sapp P E, Chin W, Geller B, Hayden D L, Schoenfeld D A, Hosler B A, Horvitz H R, Brown R H. Ann Neurol. 1997;41:210–221. - PubMed
-
- Deng H X, Hentati A, Tainer J A, Iqbal Z, Cayabyab A, Hung W Y, Getzoff E D, Hu P, Herzfeldt B, Roos R P, et al. Science. 1993;261:1047–1051. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

