In vivo immune evasion mediated by the herpes simplex virus type 1 immunoglobulin G Fc receptor
- PMID: 9620988
- PMCID: PMC110157
- DOI: 10.1128/JVI.72.7.5351-5359.1998
In vivo immune evasion mediated by the herpes simplex virus type 1 immunoglobulin G Fc receptor
Abstract
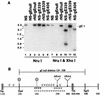
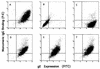
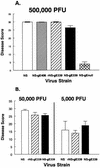
Herpes simplex virus (HSV) glycoproteins gE and gI form an immunoglobulin G (IgG) Fc receptor (FcgammaR) that binds the Fc domain of human anti-HSV IgG and inhibits Fc-mediated immune functions in vitro. gE or gI deletion mutant viruses are avirulent, probably because gE and gI are also involved in cell-to-cell spread. In an effort to modify FcgammaR activity without affecting other gE functions, we constructed a mutant virus, NS-gE339, that has four amino acids inserted into gE within the domain homologous to mammalian IgG FcgammaRs. NS-gE339 expresses gE and gI, is FcgammaR-, and does not participate in antibody bipolar bridging since it does not block activities mediated by the Fc domain of anti-HSV IgG. In vivo studies were performed with mice because the HSV-1 FcgammaR does not bind murine IgG; therefore, the absence of an FcgammaR should not affect virulence in mice. NS-gE339 causes disease at the skin inoculation site comparably to wild-type and rescued viruses, indicating that the FcgammaR- mutant virus is pathogenic in animals. Mice were passively immunized with human anti-HSV IgG and then infected with mutant or wild-type virus. We postulated that the HSV-1 FcgammaR should protect wild-type virus from antibody attack. Human anti-HSV IgG greatly reduced viral titers and disease severity in NS-gE339-infected animals while having little effect on wild-type or rescued virus. We conclude that the HSV-1 FcgammaR enables the virus to evade antibody attack in vivo, which likely explains why antibodies are relatively ineffective against HSV infection.
Figures



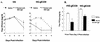
Similar articles
-
The herpes simplex virus 1 IgG fc receptor blocks antibody-mediated complement activation and antibody-dependent cellular cytotoxicity in vivo.J Virol. 2011 Apr;85(7):3239-49. doi: 10.1128/JVI.02509-10. Epub 2011 Jan 12. J Virol. 2011. PMID: 21228231 Free PMC article.
-
Immunization strategies to block the herpes simplex virus type 1 immunoglobulin G Fc receptor.J Virol. 2004 Mar;78(5):2562-71. doi: 10.1128/jvi.78.5.2562-2571.2004. J Virol. 2004. PMID: 14963159 Free PMC article.
-
Herpes simplex virus glycoproteins E and I facilitate cell-to-cell spread in vivo and across junctions of cultured cells.J Virol. 1994 Feb;68(2):834-45. doi: 10.1128/JVI.68.2.834-845.1994. J Virol. 1994. PMID: 8289387 Free PMC article.
-
Viral interference with antibody and complement.Semin Cell Dev Biol. 1998 Jun;9(3):329-37. doi: 10.1006/scdb.1998.0242. Semin Cell Dev Biol. 1998. PMID: 9665870 Free PMC article. Review.
-
Anti-retroviral antibody FcγR-mediated effector functions.Immunol Rev. 2017 Jan;275(1):285-295. doi: 10.1111/imr.12482. Immunol Rev. 2017. PMID: 28133801 Free PMC article. Review.
Cited by
-
Live attenuated herpes simplex virus 2 glycoprotein E deletion mutant as a vaccine candidate defective in neuronal spread.J Virol. 2012 Apr;86(8):4586-98. doi: 10.1128/JVI.07203-11. Epub 2012 Feb 8. J Virol. 2012. PMID: 22318147 Free PMC article.
-
Type I interferons and herpes simplex virus infection: a naked DNA approach as a therapeutic option?Immunol Res. 2001;24(1):1-11. doi: 10.1385/IR:24:1:01. Immunol Res. 2001. PMID: 11485206 Review.
-
Herpes simplex virus vectors elicit durable immune responses in the presence of preexisting host immunity.J Virol. 2002 Apr;76(8):3678-87. doi: 10.1128/jvi.76.8.3678-3687.2002. J Virol. 2002. PMID: 11907207 Free PMC article.
-
Recent progress in herpes simplex virus immunobiology and vaccine research.Clin Microbiol Rev. 2003 Jan;16(1):96-113. doi: 10.1128/CMR.16.1.96-113.2003. Clin Microbiol Rev. 2003. PMID: 12525427 Free PMC article. Review.
-
Blocking herpes simplex virus 2 glycoprotein E immune evasion as an approach to enhance efficacy of a trivalent subunit antigen vaccine for genital herpes.J Virol. 2014 Aug;88(15):8421-32. doi: 10.1128/JVI.01130-14. Epub 2014 May 14. J Virol. 2014. PMID: 24829358 Free PMC article.
References
-
- Balan P, Davis-Poynter N, Bell S, Atkinson H, Browne H, Minson T. An analysis of the in vitro and in vivo phenotypes of mutants of herpes simplex virus type 1 lacking glycoproteins gG, gE, gI or putative gJ. J Gen Virol. 1994;75:1245–1258. - PubMed
-
- Basu S, Dubin G, Basu M, Nguyen V, Friedman H M. Characterization of regions of herpes simplex virus type 1 glycoprotein E involved in binding the Fc domain of monomeric IgG and in forming a complex with glycoprotein I. J Immunol. 1995;154:260–267. - PubMed
-
- Basu S, Dubin G, Nagashunmugam T, Basu M, Goldstein L T, Wang L, Weeks B, Friedman H M. Mapping regions of herpes simplex virus type 1 glycoprotein I required for formation of the viral Fc receptor for monomeric IgG. J Immunol. 1997;158:209–215. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

