T-Cell response to woodchuck hepatitis virus (WHV) antigens during acute self-limited WHV infection and convalescence and after viral challenge
- PMID: 9621072
- PMCID: PMC110414
- DOI: 10.1128/JVI.72.7.6083-6091.1998
T-Cell response to woodchuck hepatitis virus (WHV) antigens during acute self-limited WHV infection and convalescence and after viral challenge
Abstract
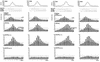
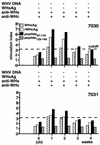
The infection of woodchucks with woodchuck hepatitis virus (WHV) provides an experimental model to study early immune responses during hepadnavirus infection that cannot be tested in patients. The T-cell response of experimentally WHV-infected woodchucks to WHsAg, rWHcAg, and WHcAg peptides was monitored by observing 5-bromo-2'-deoxyuridine and [2-3H]adenine incorporation. The first T-cell responses were directed against WHsAg 3 weeks after infection; these were followed by responses to rWHcAg including the immunodominant T-cell epitope of WHcAg (amino acids 97 to 110). Maximal proliferative responses were detected when the animals seroconvered to anti-WHs and anti-WHc (week 6). A decrease in the T-cell response to viral antigens coincided with clearance of viral DNA. Polyclonal rWHcAg-specific T-cell lines were established 6, 12, 18, and 24 weeks postinfection, and their responses to WHcAg peptides were assessed. Five to seven peptides including the immunodominant epitope were recognized throughout the observation period (6 months). At 12 months after infection, T-cell responses to antigens and peptides were not detected. Reactivation of T-cell responses to viral antigens and peptides occurred within 7 days after challenge of animals with WHV. These results demonstrate that a fast and vigorous T-cell response to WHsAg, rWHcAg, and amino acids 97 to 110 of the WHcAg occurs within 3 weeks after WHV infection. The peak of this response was associated with viral clearance and may be crucial for recovery from infection. One year after infection, no proliferation of T cells in response to antigens was observed; however, the WHV-specific T-cell response was reactivated after challenge of woodchucks with WHV and may be responsible for protection against WHV reinfection.
Figures



Similar articles
-
Characterization of T-cell response to woodchuck hepatitis virus core protein and protection of woodchucks from infection by immunization with peptides containing a T-cell epitope.J Virol. 1997 Jan;71(1):65-74. doi: 10.1128/JVI.71.1.65-74.1997. J Virol. 1997. PMID: 8985324 Free PMC article.
-
Immunization of woodchucks with plasmids expressing woodchuck hepatitis virus (WHV) core antigen and surface antigen suppresses WHV infection.J Virol. 1999 Jan;73(1):281-9. doi: 10.1128/JVI.73.1.281-289.1999. J Virol. 1999. PMID: 9847331 Free PMC article.
-
Acute resolving woodchuck hepatitis virus (WHV) infection is associated with a strong cytotoxic T-lymphocyte response to a single WHV core peptide.J Virol. 2007 Jul;81(13):7156-63. doi: 10.1128/JVI.02711-06. Epub 2007 Apr 25. J Virol. 2007. PMID: 17459928 Free PMC article.
-
Evaluation of new approaches to prophylactic and therapeutic vaccinations against hepatitis B viruses in the woodchuck model.Intervirology. 2001;44(2-3):124-31. doi: 10.1159/000050039. Intervirology. 2001. PMID: 11509873 Review.
-
Diverse Virus and Host-Dependent Mechanisms Influence the Systemic and Intrahepatic Immune Responses in the Woodchuck Model of Hepatitis B.Front Immunol. 2020 May 27;11:853. doi: 10.3389/fimmu.2020.00853. eCollection 2020. Front Immunol. 2020. PMID: 32536912 Free PMC article. Review.
Cited by
-
Electroporation enhances immunogenicity of a DNA vaccine expressing woodchuck hepatitis virus surface antigen in woodchucks.J Virol. 2011 May;85(10):4853-62. doi: 10.1128/JVI.02437-10. Epub 2011 Mar 9. J Virol. 2011. PMID: 21389124 Free PMC article.
-
Apoptosis and regeneration of hepatocytes during recovery from transient hepadnavirus infections.J Virol. 2000 Feb;74(3):1495-505. doi: 10.1128/jvi.74.3.1495-1505.2000. J Virol. 2000. PMID: 10627561 Free PMC article.
-
Recent Drug Development in the Woodchuck Model of Chronic Hepatitis B.Viruses. 2022 Aug 3;14(8):1711. doi: 10.3390/v14081711. Viruses. 2022. PMID: 36016334 Free PMC article. Review.
-
Woodchuck gamma interferon upregulates major histocompatibility complex class I transcription but is unable to deplete woodchuck hepatitis virus replication intermediates and RNAs in persistently infected woodchuck primary hepatocytes.J Virol. 2002 Jan;76(1):58-67. doi: 10.1128/jvi.76.1.58-67.2002. J Virol. 2002. PMID: 11739671 Free PMC article.
-
Needle-free, spirulina-produced Plasmodium falciparum circumsporozoite vaccination provides sterile protection against pre-erythrocytic malaria in mice.NPJ Vaccines. 2022 Oct 4;7(1):113. doi: 10.1038/s41541-022-00534-5. NPJ Vaccines. 2022. PMID: 36195607 Free PMC article.
References
-
- Akbar A N, Salmon M, Savill J, Janossy G. A possible role for bcl-2 in regulating T-cell memory: a “balancing act” between cell death and survival. Immunol Today. 1993;14:526–532. - PubMed
-
- Asada M, Galambos J T. Sorbitol dehydrogenase and hepatocellular injury: an experimental and clinical study. Gastroenterology. 1963;44:578–584.
-
- Bertoletti A, Sette A, Chisari F V, Penna A, Levrero M, De Carli M, Fiaccadori F, Ferrari C. Natural variants of cytotoxic epitopes are T-cell receptor antagonists for antiviral cytotoxic T-cells. Nature (London) 1994;369:407–410. - PubMed
-
- Buendia M A. Hepatitis B viruses and liver cancer: the woodchuck model. Symp Soc Gen Microbiol. 1994;51:183–187.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

