Phenotypic study of resistance of beta-lactamase-inhibitor-resistant TEM enzymes which differ by naturally occurring variations and by site-directed substitution at Asp276
- PMID: 9624468
- PMCID: PMC105596
- DOI: 10.1128/AAC.42.6.1323
Phenotypic study of resistance of beta-lactamase-inhibitor-resistant TEM enzymes which differ by naturally occurring variations and by site-directed substitution at Asp276
Abstract
At this time an amino acid substitution at position 276 in the TEM-1 enzyme is associated with an additional substitution at position 69 in natural beta-lactamase-inhibitor-resistant (IRT) beta-lactamases. The effect of the Asn276-->Asp substitution on resistance was assessed with the Asn276Asp variant, generated by site-directed mutagenesis. The mutant was resistant to beta-lactamase inhibitors, but the MICs of amoxicillin combined with clavulanic acid or tazobactam were strikingly different for E. coli strains producing the Asn276Asp variant and those producing naturally occurring IRTs with single or double substitutions. The inhibitory effects of clavulanic acid and tazobactam were the same in IRTs with substitutions at position 69 (IRT-5 and IRT-6). The effect of clavulanic acid on the MICs of amoxicillin for the Asn276Asp variant was greater than that of tazobactam. In IRTs with double substitutions, at positions 69 plus 276 (IRT-4, IRT-7, and IRT-8) or 69 plus 275 (IRT-14), tazobactam was a more potent inhibitor than clavulanic acid. The effect of the Asn276-->Asp substitution on the values of the kinetic constants and the concentration required to inhibit by 50% the hydrolysis of benzylpenicillin confirms that this single mutation is responsible for resistance to beta-lactamase inhibitors. Molecular modeling of the Asn276Asp mutant shows that Asp276 can form two salt bonds with Arg244 close to the penicillin-binding cavity. The addition of the Asp276 mutation to that preexisting at position 69 confers a higher selective advantage to bacteria, as shown by the reduction in beta-lactamase inhibitor efficiencies of the double variants. Therefore, the emergence of multiple mutations in TEM beta-lactamases by virtue of the use of beta-lactamase inhibitors increases selection pressure resulting in the convergent evolution of resistant strains.
Figures

 , amoxicillin;
, amoxicillin;
 , amoxicillin plus clavulanate;
□, amoxicillin plus tazobactam.
, amoxicillin plus clavulanate;
□, amoxicillin plus tazobactam.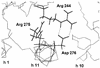
Similar articles
-
X-ray structure of the Asn276Asp variant of the Escherichia coli TEM-1 beta-lactamase: direct observation of electrostatic modulation in resistance to inactivation by clavulanic acid.Biochemistry. 1999 Jul 27;38(30):9570-6. doi: 10.1021/bi990758z. Biochemistry. 1999. PMID: 10423234
-
Aspartic acid for asparagine substitution at position 276 reduces susceptibility to mechanism-based inhibitors in SHV-1 and SHV-5 beta-lactamases.J Antimicrob Chemother. 1999 Jan;43(1):23-9. doi: 10.1093/jac/43.1.23. J Antimicrob Chemother. 1999. PMID: 10381097
-
Selection and characterization of beta-lactam-beta-lactamase inactivator-resistant mutants following PCR mutagenesis of the TEM-1 beta-lactamase gene.Antimicrob Agents Chemother. 1998 Jul;42(7):1542-8. doi: 10.1128/AAC.42.7.1542. Antimicrob Agents Chemother. 1998. PMID: 9660980 Free PMC article.
-
Class A beta-lactamases--enzyme-inhibitor interactions and resistance.Pharmacol Ther. 1999 Aug;83(2):141-51. doi: 10.1016/s0163-7258(99)00027-3. Pharmacol Ther. 1999. PMID: 10511459 Review.
-
Inhibitor-resistant TEM beta-lactamases: phenotypic, genetic and biochemical characteristics.J Antimicrob Chemother. 1999 Apr;43(4):447-58. doi: 10.1093/jac/43.4.447. J Antimicrob Chemother. 1999. PMID: 10350372 Review.
Cited by
-
The role of a second-shell residue in modifying substrate and inhibitor interactions in the SHV beta-lactamase: a study of ambler position Asn276.Biochemistry. 2009 Jun 2;48(21):4557-66. doi: 10.1021/bi9003292. Biochemistry. 2009. PMID: 19351161 Free PMC article.
-
Genetic and structural characterization of an L201P global suppressor substitution in TEM-1 beta-lactamase.J Mol Biol. 2008 Dec 5;384(1):151-64. doi: 10.1016/j.jmb.2008.09.009. Epub 2008 Sep 16. J Mol Biol. 2008. PMID: 18822298 Free PMC article.
-
Multiple global suppressors of protein stability defects facilitate the evolution of extended-spectrum TEM β-lactamases.J Mol Biol. 2010 Dec 17;404(5):832-46. doi: 10.1016/j.jmb.2010.10.008. Epub 2010 Oct 16. J Mol Biol. 2010. PMID: 20955714 Free PMC article.
-
Genetic characterization of resistance to extended-spectrum beta-lactams in Klebsiella oxytoca isolates recovered from patients with septicemia at hospitals in the Stockholm area.Antimicrob Agents Chemother. 1999 May;43(5):1294-7. doi: 10.1128/AAC.43.5.1294. Antimicrob Agents Chemother. 1999. PMID: 10223957 Free PMC article.
-
Construction and characterization of mutants of the TEM-1 beta-lactamase containing amino acid substitutions associated with both extended-spectrum resistance and resistance to beta-lactamase inhibitors.Antimicrob Agents Chemother. 1999 Aug;43(8):1881-7. doi: 10.1128/AAC.43.8.1881. Antimicrob Agents Chemother. 1999. PMID: 10428907 Free PMC article.
References
-
- Belaaouaj A, Lapoumeroulie C, Caniça M M, Vedel G, Névot P, Krishnamoorthy R, Paul G. Nucleotide sequences of the genes coding for the TEM-like β-lactamases IRT-1 and IRT-2 (formerly called TRI-1 and TRI-2) FEMS Microbiol Lett. 1994;120:75–80. - PubMed
-
- Bonomo R A, Dawes C G, Knox J R, Shlaes D M. β-Lactamase mutations far from the active site influence inhibitor binding. Biochim Biophys Acta. 1995;1247:121–125. - PubMed
-
- Bret L, Chanal C, Sirot D, Labia R, Sirot J. Characterization of an inhibitor-resistant enzyme IRT-2 derived from TEM-2 β-lactamase produced by Proteus mirabilisstrains. J Antimicrob Chemother. 1996;38:183–191. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

