Inhibition of clathrin-mediated endocytosis selectively attenuates specific insulin receptor signal transduction pathways
- PMID: 9632770
- PMCID: PMC108970
- DOI: 10.1128/MCB.18.7.3862
Inhibition of clathrin-mediated endocytosis selectively attenuates specific insulin receptor signal transduction pathways
Abstract
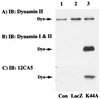
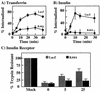
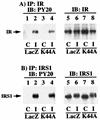
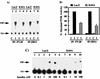
To examine the role of clathrin-dependent insulin receptor internalization in insulin-stimulated signal transduction events, we expressed a dominant-interfering mutant of dynamin (K44A/dynamin) by using a recombinant adenovirus in the H4IIE hepatoma and 3T3L1 adipocyte cell lines. Expression of K44A/dynamin inhibited endocytosis of the insulin receptor as determined by both cell surface radioligand binding and trypsin protection analysis. The inhibition of the insulin receptor endocytosis had no effect on either the extent of insulin receptor autophosphorylation or insulin receptor substrate 1 (IRS1) tyrosine phosphorylation. In contrast, expression of K44A/dynamin partially inhibited insulin-stimulated Shc tyrosine phosphorylation and activation of the mitogen-activated protein kinases ERK1 and -2. Although there was an approximately 50% decrease in the insulin-stimulated activation of the phosphatidylinositol 3-kinase associated with IRS1, insulin-stimulated Akt kinase phosphorylation and activation were unaffected. The expression of K44A/dynamin increased the basal rate of amino acid transport, which was additive with the effect of insulin but had no effect on the basal or insulin-stimulated DNA synthesis. In 3T3L1 adipocytes, expression of K44A/dynamin increased the basal rate of glucose uptake, glycogen synthesis, and lipogenesis without any significant effect on insulin stimulation. Together, these data demonstrate that the acute actions of insulin are largely independent of insulin receptor endocytosis and are initiated by activation of the plasma membrane-localized insulin receptor.
Figures



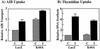
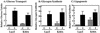
References
-
- Backer J M, Kahn C R, Cahill D A, Ullrich A, White M F. Receptor-mediated internalization of insulin requires a 12-amino acid sequence in the juxtamembrane region of the insulin receptor beta-subunit. J Biol Chem. 1990;265:16450–16454. - PubMed
-
- Backer J M, Kahn C R, White M F. Tyrosine phosphorylation of the insulin receptor during insulin-stimulated internalization in rat hepatoma cells. J Biol Chem. 1989;264:1694–1701. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
