Targeting the IL-15 receptor with an antagonist IL-15 mutant/Fc gamma2a protein blocks delayed-type hypersensitivity
- PMID: 9637483
- PMCID: PMC3806316
Targeting the IL-15 receptor with an antagonist IL-15 mutant/Fc gamma2a protein blocks delayed-type hypersensitivity
Abstract
Owing to shared receptor components, the biologic activities of IL-15 are similar to those of IL-2. However, the patterns of tissue expression of IL-2/IL-2R alpha and IL-15/IL-15R alpha differ. The development of agents targeting the receptor and signaling elements of IL-15 may provide a new perspective for treatment of diseases associated with expression of IL-15/IL-15R. We designed, genetically constructed, and expressed a receptor site-specific IL-15 antagonist by mutating glutamine residues within the C terminus of IL-15 to aspartic acid and genetically linked this mutant IL-15 to murine Fc gamma2a. These mutant IL-15 proteins specifically bind to the IL-15R, competitively inhibit IL-15-triggered cell proliferation, and do not activate the STAT-signaling pathway. Because the receptor site-specific antagonist IL-15 mutant/Fc gamma2a fusion proteins had a prolonged t(1/2) in vivo and the potential for destruction of IL-15R+ leukocytes, we examined the immunosuppressive activity of this agent. An IL-15 mutant/Fc gamma2a fusion protein markedly attenuated Ag-specific delayed-type hypersensitivity responses and decreased leukocyte infiltration within the delayed-type hypersensitivity sites. These findings suggest that 1) IL-15/IL-15R+ cells are crucial to these T cell-dependent immune responses, and 2) treatment with IL-15 mutant/Fc gamma2a protein may ameliorate T cell-dependent immune/inflammatory diseases.
Figures
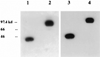


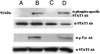



Similar articles
-
Mast cells express novel functional IL-15 receptor alpha isoforms.J Immunol. 2003 May 15;170(10):5045-55. doi: 10.4049/jimmunol.170.10.5045. J Immunol. 2003. Retraction in: J Immunol. 2011 Feb 15;186(4):2682. doi: 10.4049/jimmunol.1090143. PMID: 12734349 Retracted.
-
Targeting IL-15 receptor-bearing cells with an antagonist mutant IL-15/Fc protein prevents disease development and progression in murine collagen-induced arthritis.J Immunol. 2004 Nov 1;173(9):5818-26. doi: 10.4049/jimmunol.173.9.5818. J Immunol. 2004. PMID: 15494535
-
An antagonist IL-15/Fc protein prevents costimulation blockade-resistant rejection.J Immunol. 2001 Sep 15;167(6):3478-85. doi: 10.4049/jimmunol.167.6.3478. J Immunol. 2001. PMID: 11544341 Free PMC article.
-
The roles of interleukin-15 receptor alpha: trans-presentation, receptor component, or both?Int J Biochem Cell Biol. 2005 Aug;37(8):1567-71. doi: 10.1016/j.biocel.2005.02.017. Epub 2005 Mar 5. Int J Biochem Cell Biol. 2005. PMID: 15896666 Review.
-
Interleukin 15 and its receptor.Ciba Found Symp. 1995;195:221-9; discussion 229-33. doi: 10.1002/9780470514849.ch15. Ciba Found Symp. 1995. PMID: 8724840 Review.
Cited by
-
Addition of an IL-15 mutant/FCgamma2A antagonist protein protects islet allografts from rejection overriding costimulation blockade.Transplant Proc. 2002 May;34(3):745-7. doi: 10.1016/s0041-1345(01)02900-1. Transplant Proc. 2002. PMID: 12034169 Free PMC article. No abstract available.
-
Configuration-dependent Presentation of Multivalent IL-15:IL-15Rα Enhances the Antigen-specific T Cell Response and Anti-tumor Immunity.J Biol Chem. 2016 Apr 22;291(17):8931-50. doi: 10.1074/jbc.M115.695304. Epub 2015 Dec 30. J Biol Chem. 2016. PMID: 26719339 Free PMC article.
-
Effects of an agonist interleukin-2/Fc fusion protein, a mutant antagonist interleukin-15/Fc fusion protein, and sirolimus on cardiac allograft survival in non-human primates.J Heart Lung Transplant. 2012 Apr;31(4):427-35. doi: 10.1016/j.healun.2012.01.864. Epub 2012 Feb 24. J Heart Lung Transplant. 2012. PMID: 22366291 Free PMC article.
-
IL-15 expression on RA synovial fibroblasts promotes B cell survival.PLoS One. 2012;7(7):e40620. doi: 10.1371/journal.pone.0040620. Epub 2012 Jul 9. PLoS One. 2012. PMID: 22792388 Free PMC article.
-
Immune-Mediated Systemic Vasculitis as the Proposed Cause of Sudden-Onset Sensorineural Hearing Loss following Lassa Virus Exposure in Cynomolgus Macaques.mBio. 2018 Oct 30;9(5):e01896-18. doi: 10.1128/mBio.01896-18. mBio. 2018. PMID: 30377282 Free PMC article.
References
-
- Finkelman FD, Maliszewski C. Enhancing cytokine effects in vivo. Immunologist. 1997;5:10.
-
- Bamford R, Grant A, Burton J, Peters C, Kurys G, Goldman C, Brennan J, Roessler E, Waldmann T. The interleukin (IL) 2 receptor β chain is shared by IL-2 and a cytokine, provisionally designated IL-T, that stimulates T-cell proliferation and the induction of lymphokine-activated killer cells. Proc. Natl. Acad. Sci. USA. 1994;91:4940. - PMC - PubMed
-
- Bamford RN, Tagaya Y, Waldman TA. Interleukin 15: what it does and how it is controlled. Immunologist. 1997;5:52.
-
- Giri JG, Anderson DM, Kumaki S, Park LS, Grabstein KH, Cosman D. IL-15, a novel T cell growth factor that shares activities and receptor components with IL-2. J. Leukocyte Biol. 1995;57:763. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
