Proteasomal regulation of nuclear receptor corepressor-mediated repression
- PMID: 9637679
- PMCID: PMC316907
- DOI: 10.1101/gad.12.12.1775
Proteasomal regulation of nuclear receptor corepressor-mediated repression
Abstract
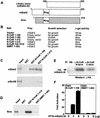
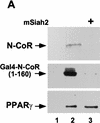
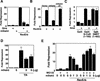
Repression of gene transcription is a fundamental property of nuclear hormone receptors. We report here that cell-specific repression by nuclear receptors correlates with levels of nuclear receptor corepressor (N-CoR) protein. N-CoR protein levels are regulated by mSiah2, a mammalian homolog of Drosophila Seven in absentia that targets N-CoR for proteasomal degradation. mSiah2 expression is cell-type specific and differentially regulates the repressive activities of nuclear receptors. These findings establish targeted proteolysis of transcriptional coregulators as a mechanism for cell-specific regulation of gene transcription.
Figures




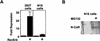
References
-
- Alland L, Muhle R, Hou H, Potes J, Chin L, Schreiber-Agus N, DePinho RA. Role for N-CoR and histone deacetylase in Sin3-mediated transcriptional repression. Nature. 1997;387:49–55. - PubMed
-
- Carthew RW, Rubin GM. seven in absentia, a gene required for specification of R7 cell fate in the Drosophila eye. Cell. 1990;63:561–577. - PubMed
-
- Chang HC, Solomon NM, Wassarman DA, Karim FD, Therrien M, Rubin GM, Wolff T. phyllopod functions in the fate determination of a subset of photoreceptors in Drosophila. Cell. 1995;80:463–472. - PubMed
-
- Chen JD, Evans RM. A transcriptional co-repressor that interacts with nuclear hormone receptors. Nature. 1995;377:454–457. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
