Renal agenesis in mice homozygous for a gene trap mutation in the gene encoding heparan sulfate 2-sulfotransferase
- PMID: 9637690
- PMCID: PMC316906
- DOI: 10.1101/gad.12.12.1894
Renal agenesis in mice homozygous for a gene trap mutation in the gene encoding heparan sulfate 2-sulfotransferase
Abstract
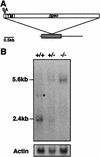
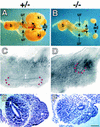
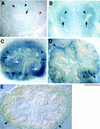
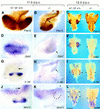
Heparan sulfate proteoglycans have been implicated in the presentation of a number of secreted signaling molecules to their signal-transducing receptors. We have characterized a gene trap mutation in the gene encoding a heparan sulfate biosynthetic enzyme, heparan sulfate 2-sulfotransferase (HS2ST). Transgenic mice were generated from embryonic stem cells harboring this insertion. lacZ reporter gene activity in heterozygous embryos demonstrates that the gene is expressed differentially during embryogenesis, presumably directing dynamic changes in heparan sulfate structure. Moreover, mice homozygous for the Hs2st gene trap allele die in the neonatal period, exhibiting bilateral renal agenesis and defects of the eye and the skeleton. Analysis of kidney development in Hs2st mutants reveals that the gene is not required for two early events-ureteric bud outgrowth from the Wolffian duct and initial induction of Pax-2 expression in the metanephric mesenchyme. It is required, however, for mesenchymal condensation around the ureteric bud and initiation of branching morphogenesis. Because 2-O-sulfation has been shown to influence the functional interactions of ligands with heparan sulfate in vitro, we discuss the possibility that the Hs2st mutant phenotype is a consequence of compromised interactions between growth factors and their signal-transducing receptors. These data provide the first genetic evidence that the regulated synthesis of differentially glycosylated proteoglycans can affect morphogenesis during vertebrate development.
Figures



Similar articles
-
Hs2st mediated kidney mesenchyme induction regulates early ureteric bud branching.Dev Biol. 2010 Mar 15;339(2):354-65. doi: 10.1016/j.ydbio.2009.12.033. Epub 2010 Jan 6. Dev Biol. 2010. PMID: 20059993 Free PMC article.
-
Maintenance of chondroitin sulfation balance by chondroitin-4-sulfotransferase 1 is required for chondrocyte development and growth factor signaling during cartilage morphogenesis.Development. 2005 Sep;132(17):3989-4003. doi: 10.1242/dev.01948. Epub 2005 Aug 3. Development. 2005. PMID: 16079159
-
Heparan sulphation patterns generated by specific heparan sulfotransferase enzymes direct distinct aspects of retinal axon guidance at the optic chiasm.J Neurosci. 2006 Jun 28;26(26):6911-23. doi: 10.1523/JNEUROSCI.0505-06.2006. J Neurosci. 2006. PMID: 16807321 Free PMC article.
-
Heparan sulfate 2-O-sulfotransferase (Hs2st) and mouse development.Glycoconj J. 2002 May-Jun;19(4-5):347-54. doi: 10.1023/A:1025325222530. Glycoconj J. 2002. PMID: 12975615 Review.
-
Growth factor-heparan sulfate "switches" regulating stages of branching morphogenesis.Pediatr Nephrol. 2014 Apr;29(4):727-35. doi: 10.1007/s00467-013-2725-z. Epub 2014 Feb 2. Pediatr Nephrol. 2014. PMID: 24488503 Review.
Cited by
-
2- and 6-O-sulfated proteoglycans have distinct and complementary roles in cranial axon guidance and motor neuron migration.Development. 2016 Jun 1;143(11):1907-13. doi: 10.1242/dev.126854. Epub 2016 Apr 5. Development. 2016. PMID: 27048738 Free PMC article.
-
Antiresorptive activity of osteoprotegerin requires an intact heparan sulfate-binding site.Proc Natl Acad Sci U S A. 2020 Jul 21;117(29):17187-17194. doi: 10.1073/pnas.2005859117. Epub 2020 Jul 7. Proc Natl Acad Sci U S A. 2020. PMID: 32636266 Free PMC article.
-
HS2ST1-dependent signaling pathways determine breast cancer cell viability, matrix interactions, and invasive behavior.Cancer Sci. 2020 Aug;111(8):2907-2922. doi: 10.1111/cas.14539. Epub 2020 Jul 9. Cancer Sci. 2020. PMID: 32573871 Free PMC article.
-
Identification of mono- and disulfated N-acetyl-lactosaminyl Oligosaccharide structures as epitopes specifically recognized by humanized monoclonal antibody HMOCC-1 raised against ovarian cancer.J Biol Chem. 2012 Feb 24;287(9):6592-602. doi: 10.1074/jbc.M111.305334. Epub 2011 Dec 22. J Biol Chem. 2012. PMID: 22194598 Free PMC article.
-
Spatiotemporal distribution of heparan sulfate epitopes during murine cartilage growth plate development.Histochem Cell Biol. 2006 Dec;126(6):713-22. doi: 10.1007/s00418-006-0203-4. Epub 2006 Jul 12. Histochem Cell Biol. 2006. PMID: 16835755
References
-
- Altschul SL, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol. 1990;215:403–410. - PubMed
-
- Bai X, Esko JD. An animal cell mutant defective in heparan sulfate hexuronic acid 2-O-sulfation. J Biol Chem. 1996;271:17711–17717. - PubMed
-
- Bai X, Bame KJ, Habuchi H, Kimata K, Esko JD. Turnover of heparan sulfate depends on 2-O-sulfation of uronic acids. J Biol Chem. 1997;272:23172–23179. - PubMed
-
- Bard JBL, Davies JA, Karavanova I, Lehtonen E, Sariola H, Vainio S. Kidney development: The inductive interactions. Sem Cell Dev Biol. 1996;7:195–202.
-
- Beddington RSP, Morgenstern J, Land H, Hogan A. An in situ transgenic enzyme marker for the midgestation mouse embryo and the visualization of inner cell mass clones during early organogenesis. Development. 1989;106:37–46. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
