Electrophysiological characterization of a putative supporting cell isolated from the frog taste disk
- PMID: 9651197
- PMCID: PMC6793497
- DOI: 10.1523/JNEUROSCI.18-14-05136.1998
Electrophysiological characterization of a putative supporting cell isolated from the frog taste disk
Abstract
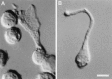
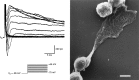
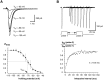
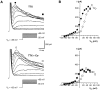
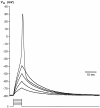
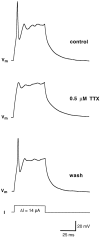
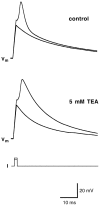
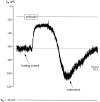
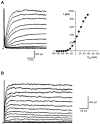
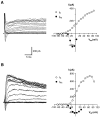
Chemosensory cells in vertebrate taste organs have two obvious specializations: an apical membrane with access to the tastants occurring in food, and synapses with sensory axons. In many species, however, certain differentiated taste cells have access to the tastants but lack any synaptic contacts with axons, and a supportive rather than chemosensory function has been attributed to them. Until now, no functional data are available for these taste cells. To begin to understand their role in taste organ physiology, we have characterized with patch-clamp recording techniques the electrophysiological properties of a putative supporting cell-the so-called wing cell-isolated from frog taste disks. Wing cells were distinguished from chemosensory elements by the presence of a typical, sheet-like apical process. Their resting potential was approximately -52 mV, and the average input resistance was 4.8 GOmega. Wing cells possessed voltage-gated Na+ currents sensitive to TTX, and an inactivating, voltage-gated K+ current sensitive to TEA. Current injections elicited single action potentials but not repetitive firing. We found no evidence for voltage-gated Ca2+ currents under various experimental conditions. Amiloride-sensitive Na+ channels, thought to be involved in Na+ chemotransduction, were present in wing cells. Many of the membrane properties of wing cells have been also reported for chemosensory taste cells. The presence of ion channels in wing cells might be suggestive of a role in controlling the microenvironment inside the taste organs or the functioning of chemosensory cells or both. In addition, they might participate directly in the sensory transduction events by allowing loop currents to flow inside the taste organs during chemostimulation.
Figures



References
-
- Akabas MH, Dodd J, Al-Awqati Q. Identification of electrophysiologically distinct subpopulations of rat taste cells. J Membr Biol. 1990;114:71–78. - PubMed
-
- Avenet P, Lindemann B. Patch-clamp study of isolated taste receptor cells of the frog. J Membr Biol. 1987;97:223–240. - PubMed
-
- Avenet P, Lindemann B. Amiloride-blockable sodium currents in isolated taste receptor cells. J Membr Biol. 1988;105:245–255. - PubMed
-
- Barres BA, Chun LLY, Corey DP. Ion channels in vertebrate glia. Annu Rev Neurosci. 1990;13:441–474. - PubMed
-
- Barry PH, Lynch JW. Liquid junction potentials and small cell effects in patch clamp analysis. J Membr Biol. 1991;121:101–117. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
