Actin filaments and microtubules are involved in different membrane traffic pathways that transport sphingolipids to the apical surface of polarized HepG2 cells
- PMID: 9658181
- PMCID: PMC25441
- DOI: 10.1091/mbc.9.7.1939
Actin filaments and microtubules are involved in different membrane traffic pathways that transport sphingolipids to the apical surface of polarized HepG2 cells
Abstract
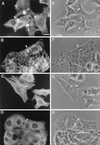
In polarized HepG2 hepatoma cells, sphingolipids are transported to the apical, bile canalicular membrane by two different transport routes, as revealed with fluorescently tagged sphingolipid analogs. One route involves direct, transcytosis-independent transport of Golgi-derived glucosylceramide and sphingomyelin, whereas the other involves basolateral to apical transcytosis of both sphingolipids. We show that these distinct routes display a different sensitivity toward nocodazole and cytochalasin D, implying a specific transport dependence on either microtubules or actin filaments, respectively. Thus, nocodazole strongly inhibited the direct route, whereas sphingolipid transport by transcytosis was hardly affected. Moreover, nocodazole blocked "hyperpolarization," i.e., the enlargement of the apical membrane surface, which is induced by treating cells with dibutyryl-cAMP. By contrast, the transcytotic route but not the direct route was inhibited by cytochalasin D. The actin-dependent step during transcytotic lipid transport probably occurs at an early endocytic event at the basolateral plasma membrane, because total lipid uptake and fluid phase endocytosis of horseradish peroxidase from this membrane were inhibited by cytochalasin D as well. In summary, the results show that the two sphingolipid transport pathways to the apical membrane must have a different requirement for cytoskeletal elements.
Figures
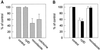

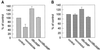
Similar articles
-
Sphingolipid transport to the apical plasma membrane domain in human hepatoma cells is controlled by PKC and PKA activity: a correlation with cell polarity in HepG2 cells.J Cell Biol. 1997 Jul 28;138(2):307-21. doi: 10.1083/jcb.138.2.307. J Cell Biol. 1997. PMID: 9230073 Free PMC article.
-
Both microtubules and actin filaments are required for efficient postendocytotic traffic of the polymeric immunoglobulin receptor in polarized Madin-Darby canine kidney cells.J Biol Chem. 1997 Mar 7;272(10):6741-51. doi: 10.1074/jbc.272.10.6741. J Biol Chem. 1997. PMID: 9045707
-
(Glyco)sphingolipids are sorted in sub-apical compartments in HepG2 cells: a role for non-Golgi-related intracellular sites in the polarized distribution of (glyco)sphingolipids.J Cell Biol. 1998 Aug 10;142(3):683-96. doi: 10.1083/jcb.142.3.683. J Cell Biol. 1998. PMID: 9700158 Free PMC article.
-
Mechanisms and functional features of polarized membrane traffic in epithelial and hepatic cells.Biochem J. 1998 Dec 1;336 ( Pt 2)(Pt 2):257-69. doi: 10.1042/bj3360257. Biochem J. 1998. PMID: 9820799 Free PMC article. Review.
-
Endocytic traffic in polarized epithelial cells: role of the actin and microtubule cytoskeleton.Traffic. 2001 Mar;2(3):149-59. doi: 10.1034/j.1600-0854.2001.020301.x. Traffic. 2001. PMID: 11260520 Review.
Cited by
-
Binding of CAP70 to inducible nitric oxide synthase and implications for the vectorial release of nitric oxide in polarized cells.Mol Biol Cell. 2007 Jul;18(7):2768-77. doi: 10.1091/mbc.e06-12-1102. Epub 2007 May 16. Mol Biol Cell. 2007. PMID: 17507652 Free PMC article.
-
Bifunctional small molecules that mediate the degradation of extracellular proteins.Nat Chem Biol. 2021 Sep;17(9):947-953. doi: 10.1038/s41589-021-00851-1. Epub 2021 Aug 19. Nat Chem Biol. 2021. PMID: 34413525
-
Uptake and transport of PEG-graft-trimethyl-chitosan copolymer-insulin nanocomplexes by epithelial cells.Pharm Res. 2005 Dec;22(12):2058-68. doi: 10.1007/s11095-005-8175-y. Epub 2005 Sep 26. Pharm Res. 2005. PMID: 16170693
-
Structure and function of ABCG2-rich extracellular vesicles mediating multidrug resistance.PLoS One. 2011 Jan 24;6(1):e16007. doi: 10.1371/journal.pone.0016007. PLoS One. 2011. PMID: 21283667 Free PMC article.
-
Polarized sphingolipid transport from the subapical compartment: evidence for distinct sphingolipid domains.Mol Biol Cell. 1999 Oct;10(10):3449-61. doi: 10.1091/mbc.10.10.3449. Mol Biol Cell. 1999. PMID: 10512879 Free PMC article.
References
-
- Babia T, Kok JW, Hoekstra D. The use of fluorescent lipid analogues to study endocytosis of glycosphingolipids. In: Greenstein B, editor. Receptor Research Methods. London: Harwood Academic; 1994. pp. 155–174.
-
- Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–917. - PubMed
-
- Boyer JL, Soroka CJ. Vesicle targeting to the apical domain regulates bile excretory function in isolated rat hepatocyte couplets. Gastroenterology. 1995;109:1600–1611. - PubMed
-
- Chazaud B, Muriel MP, Bauvy C, Codogno P, Aubery M, Decastel M. Requirement of either the NH4Cl-sensitive or the cytochalasin D-sensitive pathway for ricin toxicity depends upon the enterocytic state of differentiation of HT-29 cells. Eur J Cell Biol. 1994;64:15–28. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

