Mutual occlusion of P2X ATP receptors and nicotinic receptors on sympathetic neurons of the guinea-pig
- PMID: 9660893
- PMCID: PMC2231072
- DOI: 10.1111/j.1469-7793.1998.783bj.x
Mutual occlusion of P2X ATP receptors and nicotinic receptors on sympathetic neurons of the guinea-pig
Abstract
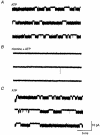
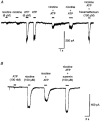
1. The interaction of ion channels activated by nicotinic receptor agonists with ion channels gated by extracellular ATP (i.e. P2X receptors) was studied on sympathetic neurons acutely dissociated from coeliac ganglia of the guinea-pig. Patch clamp methods were used to measure the inward current generated through these non-selective cationic channels under voltage clamp. 2. At the whole cell level, the specific nicotinic receptor agonists nicotine (5-100 microM) or cytisine (50-75 microM) and the P2X receptor agonists ATP (0.1-7 microM) or alpha,beta-methylene ATP (6 microM) were examined separately and in the presence of the other receptor activator. When a nicotinic and P2X receptor agonist were applied together, mutually occlusive effects were generally observed. This occurred even with concentrations of agonists that in themselves generated little to no inward current. 3. The occlusive effects of nicotinic agonists on ATP-gated currents were blocked by the nicotinic receptor/ion channel blocker hexamethonium (150 microM). The occlusive effects of ATP analogues on inward currents generated by nicotinic agonists were blocked by the P2X receptor antagonist suramin (100 microM). 4. Mutual occlusion of the effects of nicotinic agonists and ATP analogues were also observed when currents through single channels were studied in excised (outside-out) patches. 5. The results suggest that nicotinic receptors and P2X ATP receptors do not act independently in these sympathetic neurons.
Figures



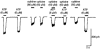
References
-
- Albuquerque EX, Alkondon M, Pereira E, Castro NG, Schrattenholz A, Barbosa TF, Bonfante-Cabarcas R, Aracava Y, Eisenberg HM, Maelicke A. Properties of neuronal nicotinic acetylcholine receptors: Pharmacological characterization and modulation of synaptic function. Journal of Pharmacology and Experimental Therapeutics. 1997;280:1117–1136. - PubMed
-
- Bean BP. Pharmacology and electrophysiology of ATP-activated ion channels. Trends in Pharmacological Sciences. 1992;13:87–90. - PubMed
-
- Brake AJ, Julius D. Signalling by extracellular nucleotides. Annual Review of Cell and Developmental Biology. 1996;12:19–41. - PubMed
-
- Buell G, Collo G, Rassendren F. P2X receptors: an emerging channel family. European Journal of Neuroscience. 1996;8:2221–2228. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

