Molecular basis for antigenic variation of a protective strain-specific antigen of Ehrlichia risticii
- PMID: 9673249
- PMCID: PMC108402
- DOI: 10.1128/IAI.66.8.3682-3688.1998
Molecular basis for antigenic variation of a protective strain-specific antigen of Ehrlichia risticii
Abstract
Ehrlichia risticii, the causative agent of Potomac horse fever, has recently been isolated from many vaccinated horses with typical clinical signs of the disease. The heterogeneity of the E. risticii isolates obtained from the vaccinated horses necessitates the identification of the molecular basis of strain variations to elucidate the vaccine failure and to aid in the development of an efficient vaccine against this disease. As an attempt, two major cross-reacting surface antigen genes of 50- and 85-kDa antigens, present separately in strains 25-D (isolated in 1984) and 90-12 (isolated in 1990 from a vaccinated horse), respectively, were cloned and sequenced. A comparative sequence analysis revealed differences and similarities between these two antigens with strain-specific sizes (SSA). The 2.5- and 1.6-kb genes coding for the 85- and 50-kDa proteins, respectively, contained many different tandem repeats. The identical repeat motifs were more frequent in the middle of both genes, but the numbers and positions of the repeats were altogether different in the genes. Many of these direct repeats of both genes had exact sequence homology and coded for the same amino acids. The homology of the 5'- and 3'-flanking regions of the two genes was greater than that of the regions in the central part of the genes. A comparative analysis of the deduced amino acid sequences of these two antigen genes indicated eight common domains, which were designated identical domains. Although the sequence homologies of these identical domains were the same, the positions of the domains in their respective strains were completely different. This finding might be one of the bases of antigenic variation between the strains. In addition, there were a few unique regions in both antigen genes where no sequence homology existed. These specific regions were designated unique domains. The 50-kDa protein had two such unique domains, and the 85-kDa protein had six such unique domains. The presence of such unique domains contributed to the large size variation of these SSA. The cross-reactivity of recombinant proteins confirmed the presence of conserved epitopes between these two antigens. The SSA have been determined to be apparent protective antigens of E. risticii.
Figures
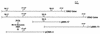



References
-
- Dutta S K, Rice R M, Hughes T D, Savage P K, Myrup A C. Detection of serum antibodies against Ehrlichia risticii in Potomac horse fever by enzyme-linked-immunosorbent assay. Vet Immunol Immunopathol. 1987;14:85–92. - PubMed
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Research Materials

