DNA end-independent activation of DNA-PK mediated via association with the DNA-binding protein C1D
- PMID: 9679063
- PMCID: PMC317006
- DOI: 10.1101/gad.12.14.2188
DNA end-independent activation of DNA-PK mediated via association with the DNA-binding protein C1D
Abstract
DNA-dependent protein kinase (DNA-PK), which is involved in DNA double-strand break repair and V(D)J recombination, is comprised of a DNA-targeting component termed Ku and an approximately 465-kD catalytic subunit, DNA-PKcs. Although DNA-PK phosphorylates proteins in the presence of DSBs or other discontinuities in the DNA double helix in vitro, the possibility exists that it is also activated in other circumstances via its association with additional proteins. Here, through use of the yeast two-hybrid screen, we discover that the recently identified high affinity DNA binding protein C1D interacts with the putative leucine zipper region of DNA-PKcs. Furthermore, we show that C1D can interact with DNA-PK in mammalian cells and that C1D is a very effective DNA-PK substrate in vitro. Finally, we establish that C1D directs the activation of DNA-PK in a manner that does not require DNA termini. Therefore, these studies provide a function for C1D and suggest novel mechanisms for DNA-PK activation in vivo.
Figures










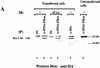
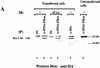
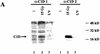
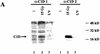
References
-
- Alt FW, Oltz EM, Young F, Gorman J, Taccioli G, Chen J. VDJ recombination. Immunol Today. 1992;13:306–314. - PubMed
-
- Anderson CW, Lees-Miller SP. The nuclear serine/threonine protein kinase DNA-PK. Crit Rev Eukaryot Gene Expr. 1992;2:283–314. - PubMed
-
- Avramova Z, Tsanev R. Stable DNA protein complexes in eukaryotic chromatin. J Mol Biol. 1987;196:437–440. - PubMed
-
- Berezney R. Organization and function of the nuclear matrix. Chrom Non-Histone Proteins. 1984;4:119–179.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
