Chloride and non-selective cation channels in unstimulated trout red blood cells
- PMID: 9679175
- PMCID: PMC2231098
- DOI: 10.1111/j.1469-7793.1998.213bi.x
Chloride and non-selective cation channels in unstimulated trout red blood cells
Abstract
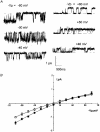
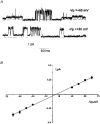
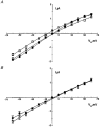
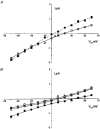
1. The cell-attached and excised inside-out configurations of the patch-clamp technique were used to demonstrate the presence of two different types of ion channels in the membrane of trout red blood cells under isotonic and normoxic conditions, in the absence of hormonal stimulation. The large majority (93%) of successful membrane seals allowed observation of at least one channel type. 2. In the cell-attached mode with Ringer solution in the bath and Ringer solution, 145 mM KCl or 145 NaCl in the pipette, a channel of intermediate conductance (15-25 pS at clamped voltage, Vp = 0 mV) was present in 85% of cells. The single channel activity reversed between 5 and 7 mV positive to the spontaneous membrane potential. A small conductance channel of 5-6 pS and +5 mV reversal potential was also present in 62% of cells. 3. After excision into the inside-out configuration (with 145 mM KCl or NaCl, pCa 8 in the bath, 145 mM KCl or NaCl, pCa 3 in the pipette) the intermediate conductance channel was present in 439 out of 452 successful seals. This channel was spontaneously active in 90% of patches and in the other 10% of patches the channel was activated by suction. The current-voltage relationship showed slight inward rectification. The channel conductance was in the range 15-20 pS between -60 and 0 mV and increased to 25-30 pS between 0 and 60 mV, with a reversal potential close to zero. Substitution of K+ for Na+ in the pipette or in the bath did not significantly change the single channel conductance. Dilution of the bathing solution KCl concentration shifted the reversal potential towards the Nernst equilibrium for cations. Substitution of N-methyl-D-glucamine (NMDG) for K+ or Na+ in the bath almost abolished the outward current whilst the divalent cation Ca2+ permeated the channel with a higher permeability than K+ and Na+. Inhibition of channel openings was obtained with flufenamic acid, quinine, gadolinium or barium. Taken together these data demonstrate that the intermediate conductance channel belongs to a class of non-selective cation (NSC) channels. 4. In excised patches, under the same control conditions, the conductance of the small conductance non-rectifying channel was 8.6 +/- 0.8 pS (n = 12) between -60 and +60 mV and the reversal potential was close to 0 mV. This channel could be blocked by 5-nitro-2-(3-phenylpropylamino)-benzoate (NPPB) but not by flufenamic acid, DIDS, barium or gadolinium. Selectivity and substitution experiments made it possible to identify this channel as a non-rectifying small conductance chloride (SCC) channel.
Figures

Similar articles
-
Characterization of large-conductance chloride channels in rabbit colonic smooth muscle.J Physiol. 1992 Mar;448:355-82. doi: 10.1113/jphysiol.1992.sp019046. J Physiol. 1992. PMID: 1375640 Free PMC article.
-
A patch-clamp study of ion channels in protoplasts prepared from the marine alga Valonia utricularis.J Membr Biol. 1999 Dec 1;172(3):235-47. doi: 10.1007/s002329900600. J Membr Biol. 1999. PMID: 10568793
-
Activation of ionic channels by deoxycholate in frog and human cell lines.Exp Physiol. 1999 May;84(3):489-99. Exp Physiol. 1999. PMID: 10362847
-
A component of platypus (Ornithorhynchus anatinus) venom forms slow-kinetic cation channels.J Membr Biol. 1999 Nov 1;172(1):37-45. doi: 10.1007/s002329900581. J Membr Biol. 1999. PMID: 10552012 Review.
-
Nonselective cation channels in cells of the crypt-base of rat distal colon.EXS. 1993;66:219-22. doi: 10.1007/978-3-0348-7327-7_17. EXS. 1993. PMID: 7505653 Review.
Cited by
-
O(2)-dependent K(+) fluxes in trout red blood cells: the nature of O(2) sensing revealed by the O(2) affinity, cooperativity and pH dependence of transport.J Physiol. 2000 Jul 1;526 Pt 1(Pt 1):69-80. doi: 10.1111/j.1469-7793.2000.t01-1-00069.x. J Physiol. 2000. PMID: 10878100 Free PMC article.
-
Inhibition of cation channels in human erythrocytes by spermine.J Membr Biol. 2010 Oct;237(2-3):93-106. doi: 10.1007/s00232-010-9310-1. Epub 2010 Nov 10. J Membr Biol. 2010. PMID: 21063869
-
Mechanosensitive cation channels in human leukaemia cells: calcium permeation and blocking effect.J Physiol. 2002 May 15;541(Pt 1):81-90. doi: 10.1113/jphysiol.2001.015222. J Physiol. 2002. PMID: 12015421 Free PMC article.
-
Hemolysis of erythrocytes by granulysin-derived peptides but not by granulysin.Antimicrob Agents Chemother. 2005 Jan;49(1):388-97. doi: 10.1128/AAC.49.1.388-397.2005. Antimicrob Agents Chemother. 2005. PMID: 15616319 Free PMC article.
References
-
- Barry PH, Lynch JW. Liquid junction potentials and small cell effects in patch-clamp analysis. Journal of Membrane Biology. 1991;12:101–117. - PubMed
-
- Bear CE. A nonselective cation channel in rat liver cells is activated by membrane stretch. American Journal of Physiology. 1990;258:C421–428. - PubMed
-
- Christensen O. Mediation of cell volume regulation by Ca2+ influx through stretch-activated channels. Nature. 1987;330:66–68. - PubMed
-
- Christophersen P. Ca-activated K channel from human erythrocyte membranes: single channel rectification and selectivity. Journal of Membrane Biology. 1991;119:75–83. - PubMed
-
- Christophersen P, Benekou P. Evidence for a voltage-gated, non-selective cation channel in the human red cell membrane. Biochimica et Biophysica Acta. 1991;1065:103–106. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

