Biosynthesis of Di-myo-inositol-1,1'-phosphate, a novel osmolyte in hyperthermophilic archaea
- PMID: 9683472
- PMCID: PMC107359
- DOI: 10.1128/JB.180.15.3785-3792.1998
Biosynthesis of Di-myo-inositol-1,1'-phosphate, a novel osmolyte in hyperthermophilic archaea
Abstract
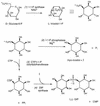
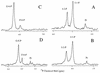
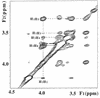
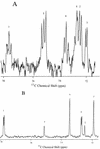
Biosynthesis of di-myo-inositol-1,1'-phosphate (DIP) is proposed to occur with myo-inositol and myo-inositol 1-phosphate (I-1-P) used as precursors. Activation of the I-1-P with CTP and condensation of the resultant CDP-inositol (CDP-I) with myo-inositol then generates DIP. The sole known biosynthetic pathway of inositol in all organisms is the conversion of D-glucose-6-phosphate to myo-inositol. This conversion requires two key enzymes: L-I-1-P synthase and I-1-P phosphatase. Enzymatic assays using 31P nuclear magnetic resonance spectroscopy as well as a colorimetric assay for inorganic phosphate have confirmed the occurrence of L-I-1-P synthase and a moderately specific I-1-P phosphatase. The enzymatic reaction that couples CDP-I with myo-inositol to generate DIP has also been detected in Methanococcus igneus. 13C labeling studies with [2,3-13C]pyruvate and [3-13C]pyruvate were used to examine this pathway in M. igneus. Label distribution in DIP was consistent with inositol units formed from glucose-6-phosphate, but the label in the glucose moiety was scrambled via transketolase and transaldolase activities of the pentose phosphate pathway.
Figures



References
-
- Chen I W, Charalampous F C. Biochemical studies on inositol. VIII. Purification and properties of the enzyme system which converts glucose-6-phosphate to inositol. J Biol Chem. 1965;240:3507–3512. - PubMed
-
- Ciulla R A. Response of methanogens to osmotic stress: in vitro and in vivo NMR studies. Ph.D. dissertation. Boston, Mass: Department of Chemistry, Boston College; 1995.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

