Small heat shock protein of Methanococcus jannaschii, a hyperthermophile
- PMID: 9689045
- PMCID: PMC21303
- DOI: 10.1073/pnas.95.16.9129
Small heat shock protein of Methanococcus jannaschii, a hyperthermophile
Abstract
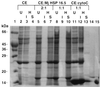
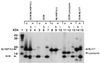
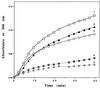
Small heat shock proteins (sHSPs) belong to a family of 12- to 43-kDa proteins that are ubiquitous and are conserved in amino acid sequence among all organisms. A sHSP homologue of Methanococcus jannaschii, a hyperthermophilic Archaeon, forms a homogeneous multimer comprised of 24 monomers with a molecular mass of 400 kDa in contrast to other sHSPs that show heterogeneous oligomeric complexes. Electron microscopy analysis revealed a spherically shaped oligomeric structure approximately 15-20 nm in diameter. The protein confers thermal protection of other proteins in vitro as found in other sHSPs. Escherichia coli cell extracts containing the protein were protected from heat-denatured precipitation when heated up to 100 degreesC, whereas extracts from cells not expressing the protein were heat-sensitive at 60 degreesC. Similar results were obtained when purified sHSP protein was added to an E. coli cell lysate. The protein also prevented the aggregation of two purified proteins: single-chain monellin (SCM) at 80 degreesC and citrate synthase at 40 degreesC.
Figures


References
-
- Gething M J, Sambrook J. Nature (London) 1992;355:33–45. - PubMed
-
- Hendrick J P, Hartl F-U. Annu Rev Biochem. 1993;62:349–384. - PubMed
-
- Parsell D A, Lindquist S. In: The Biology of Heat Shock Proteins and Molecular Chaperones. Morimoto R I, Tissieres A, Georgopoulos C, editors. Cold Spring Harbor, NY: Cold Spring Harbor Lab. Press; 1994. pp. 457–494.
-
- Buchner J. FASEB J. 1996;10:10–19. - PubMed
-
- Caspers G-J, Leunissen J A M, de Jong W W. J Mol Evol. 1995;40:238–248. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

