Modulation of human immunodeficiency virus type 1-induced syncytium formation by the conformational state of LFA-1 determined by a new luciferase-based syncytium quantitative assay
- PMID: 9696806
- PMCID: PMC109934
- DOI: 10.1128/JVI.72.9.7125-7136.1998
Modulation of human immunodeficiency virus type 1-induced syncytium formation by the conformational state of LFA-1 determined by a new luciferase-based syncytium quantitative assay
Abstract
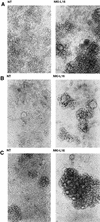
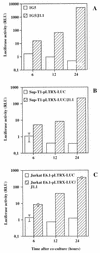
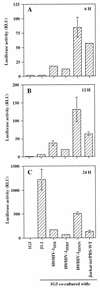
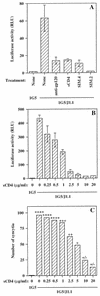
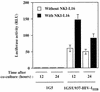
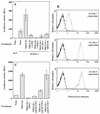
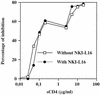
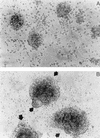
The ICAM-1/LFA-1 interaction has been clearly demonstrated to play an active role in syncytium formation induced by human immunodeficiency virus type 1 (HIV-1). Since it is known that a high-affinity state of LFA-1 for ICAM-1 can be induced through conformational change, such a high-affinity state may also contribute to the process of syncytium formation. In this study, we have investigated the involvement of the conformational status of LFA-1 in HIV-1-dependent syncytium formation by using the anti-LFA-1 antibody NKI-L16, which is known to activate the high-affinity state. Initial visual observations by light microscopy indeed suggested that the addition of the NKI-L16 antibody led to bigger and more numerous syncytia when different cell lines were tested. To further analyze this NKI-L16-dependent increment of syncytium formation in a quantitative assay, a new luciferase-based assay was developed by using a T-cell line containing an HIV-1 long terminal repeat (LTR)-driven luciferase construct (1G5) in coincubation with an HIV-1-positive cell line (J1.1). Upon fusion, the viral Tat protein could diffuse to the 1G5 cells, leading to a transcriptional increase of the HIV-1 LTR-driven luciferase gene. Initial evaluation of this assay showed a good correlation between the level of syncytium formation determined by microscopic observation and the level of measured luciferase activity. In addition, this assay showed a greater induction of enzymatic activity correlating with syncytium formation in comparison to a similar incubation with the HeLa-CD4-LTR-beta-gal indicator cell line. By using this test, NKI-L16 treatment of 1G5/J1.1 cells led to a three- to sevenfold increase in HIV-1 LTR-driven luciferase activity. The syncytium-dependent luciferase activity in NKI-L16-treated cells could be blocked by classical syncytium inhibitors such as soluble CD4, anti-CD4, and anti-gp120 antibodies. Inhibition could also be observed with specific blocking agents for the chemokine receptor CXCR4, as well as with soluble ICAM-1, anti-LFA-1, anti-ICAM-1, and anti-ICAM-2 blocking antibodies, indicating the requirement for the LFA-1/ICAM interaction. Treatment of peripheral blood mononuclear cells with NKI-L16 resulted in a higher level of syncytium formation in the presence of the cell line J1.1. Conversely, when PBMCs were infected with two different syncytium-inducing HIV-1 primary isolates, coincubation with NKI-L16-pretreated 1G5 cells led to higher levels of luciferase activity for both virus isolates. Our results therefore show for the first time a direct role for the LFA-1 high-affinity state in virus-mediated syncytium formation. Based on the demonstration that an increase in ICAM-1 binding is induced by T-cell activation, these data suggest an in vivo involvement of the high-affinity state of LFA-1 in HIV-1-induced syncytium formation. Moreover, syncytia might preferentially occur in lymph nodes, since this microenvironment harbors a high proportion of activated T cells.
Figures



References
-
- Aguilar-Cordova E, Chinen J, Donehower L, Lewis D E, Belmont J W. A sensitive reporter cell line for HIV-1 tat activity, HIV-1 inhibitors, and T cell activation effects. AIDS Res Hum Retroviruses. 1994;10:295–301. - PubMed
-
- Alkhatib G, Combadiere C, Broder C C, Feng Y, Kennedey P E, Murphy P M, Berger E A. CC CKR5: a RANTES, MIP-1α, MIP-1β receptor as a fusion cofactor for macrophage-tropic HIV-1. Science. 1996;272:1955–1959. - PubMed
-
- Allen A D, Hart D N, Hechinger M K, Slattery M J, Chesson C V, Vidikan P. Leukocyte adhesion molecules as a cofactor in AIDS: basic science and pilot study. Med Hypotheses. 1995;45:164–168. - PubMed
-
- Barbeau B, Bernier R, Dumais N, Briand G, Olivier M, Faure R, Posner B I, Tremblay M. Activation of HIV-1 LTR transcription and virus replication via NF-κB-dependent and -independent pathways by potent phosphotyrosine phosphatase inhibitors, the peroxovanadium compounds. J Biol Chem. 1997;272:12968–12977. - PubMed
-
- Bazil V, Stefanova I, Hilgert I, Kristofova H, Vanek S, Horejsi V. Monoclonal antibodies against leukocytes antigens IV. Antibodies against the LFA-1 (CD11a/CD18) leukocyte-adhesion glycoprotein. Folia Biol. 1990;36:41–50. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

