Rotavirus infection reduces sucrase-isomaltase expression in human intestinal epithelial cells by perturbing protein targeting and organization of microvillar cytoskeleton
- PMID: 9696817
- PMCID: PMC109945
- DOI: 10.1128/JVI.72.9.7228-7236.1998
Rotavirus infection reduces sucrase-isomaltase expression in human intestinal epithelial cells by perturbing protein targeting and organization of microvillar cytoskeleton
Abstract
Rotavirus infection is the most common cause of severe infantile gastroenteritis worldwide. These viruses infect mature enterocytes of the small intestine and cause structural and functional damage, including a reduction in disaccharidase activity. It was previously hypothesized that reduced disaccharidase activity resulted from the destruction of rotavirus-infected enterocytes at the villus tips. However, this pathophysiological model cannot explain situations in which low disaccharidase activity is observed when rotavirus-infected intestine exhibits few, if any, histopathologic changes. In a previous study, we demonstrated that the simian rotavirus strain RRV replicated in and was released from human enterocyte-like Caco-2 cells without cell destruction (N. Jourdan, M. Maurice, D. Delautier, A. M. Quero, A. L. Servin, and G. Trugnan, J. Virol. 71:8268-8278, 1997). In the present study, to reinvestigate disaccharidase expression during rotavirus infection, we studied sucrase-isomaltase (SI) in RRV-infected Caco-2 cells. We showed that SI activity and apical expression were specifically and selectively decreased by RRV infection without apparent cell destruction. Using pulse-chase experiments and cell surface biotinylation, we demonstrated that RRV infection did not affect SI biosynthesis, maturation, or stability but induced the blockade of SI transport to the brush border. Using confocal laser scanning microscopy, we showed that RRV infection induces important alterations of the cytoskeleton that correlate with decreased SI apical surface expression. These results lead us to propose an alternate model to explain the pathophysiology associated with rotavirus infection.
Figures

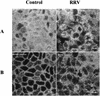
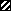 , control; □,
RRV infected. (B) At the same times p.i., infected cells were fixed
with 3% paraformaldehyde and permeabilized with Triton X-100. RRV
antigens were immunostained with a polyclonal anti-group A rotavirus
and fluorescein-labeled anti-rabbit IgG secondary antibodies (left).
Infected monolayers were also examined by phase-contrast microscopy
(right). The bar indicates 25 μm.
, control; □,
RRV infected. (B) At the same times p.i., infected cells were fixed
with 3% paraformaldehyde and permeabilized with Triton X-100. RRV
antigens were immunostained with a polyclonal anti-group A rotavirus
and fluorescein-labeled anti-rabbit IgG secondary antibodies (left).
Infected monolayers were also examined by phase-contrast microscopy
(right). The bar indicates 25 μm.


Similar articles
-
Rotavirus infection induces an increase in intracellular calcium concentration in human intestinal epithelial cells: role in microvillar actin alteration.J Virol. 2000 Mar;74(5):2323-32. doi: 10.1128/jvi.74.5.2323-2332.2000. J Virol. 2000. PMID: 10666263 Free PMC article.
-
A cyclic AMP protein kinase A-dependent mechanism by which rotavirus impairs the expression and enzyme activity of brush border-associated sucrase-isomaltase in differentiated intestinal Caco-2 cells.Cell Microbiol. 2004 Aug;6(8):719-31. doi: 10.1111/j.1462-5822.2004.00396.x. Cell Microbiol. 2004. PMID: 15236639
-
Rotavirus impairs the biosynthesis of brush-border-associated dipeptidyl peptidase IV in human enterocyte-like Caco-2/TC7 cells.Cell Microbiol. 2007 Mar;9(3):779-89. doi: 10.1111/j.1462-5822.2006.00827.x. Epub 2006 Nov 1. Cell Microbiol. 2007. PMID: 17081193
-
Biosynthesis and assembly of the largest and major intrinsic polypeptide of the small intestinal brush borders.Ciba Found Symp. 1983;95:92-112. doi: 10.1002/9780470720769.ch7. Ciba Found Symp. 1983. PMID: 6342999 Review.
-
Biosynthesis and transport of plasma membrane glycoproteins in the rat intestinal epithelial cell: studies with sucrase-isomaltase.Ciba Found Symp. 1983;95:132-63. doi: 10.1002/9780470720769.ch9. Ciba Found Symp. 1983. PMID: 6303720 Review.
Cited by
-
Rotavirus infection induces an increase in intracellular calcium concentration in human intestinal epithelial cells: role in microvillar actin alteration.J Virol. 2000 Mar;74(5):2323-32. doi: 10.1128/jvi.74.5.2323-2332.2000. J Virol. 2000. PMID: 10666263 Free PMC article.
-
Diversity in Rotavirus-Host Glycan Interactions: A "Sweet" Spectrum.Cell Mol Gastroenterol Hepatol. 2016 Mar 12;2(3):263-273. doi: 10.1016/j.jcmgh.2016.03.002. eCollection 2016 May. Cell Mol Gastroenterol Hepatol. 2016. PMID: 28090561 Free PMC article. Review.
-
Host immunity and pathogen strain contribute to intestinal disaccharidase impairment following gut infection.J Immunol. 2011 Oct 1;187(7):3769-75. doi: 10.4049/jimmunol.1100606. Epub 2011 Aug 26. J Immunol. 2011. PMID: 21873528 Free PMC article.
-
How do the rotavirus NSP4 and bacterial enterotoxins lead differently to diarrhea?Virol J. 2007 Mar 21;4:31. doi: 10.1186/1743-422X-4-31. Virol J. 2007. PMID: 17376232 Free PMC article. Review.
-
Molecular Survey of Viral and Bacterial Causes of Childhood Diarrhea in Khartoum State, Sudan.Front Microbiol. 2018 Feb 12;9:112. doi: 10.3389/fmicb.2018.00112. eCollection 2018. Front Microbiol. 2018. PMID: 29487571 Free PMC article.
References
-
- Alves de Matos A P, Carvalho Z G. African swine fever virus interaction with microtubules. Biol Cell. 1993;78:229–234. - PubMed
-
- Ball J M, Tian P, Zeng C Q-Y, Morris A P, Estes M K. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science. 1996;272:101–104. - PubMed
-
- Baricault L, Garcia M, Cibert C, Sapin C, Geraud G, Codogno P, Trugnan G. Forskolin blocks the apical expression of dipeptidyl peptidase IV in Caco-2 cells and induces its retention in lamp-1-containing vesicles. Exp Cell Res. 1993;209:277–287. - PubMed
-
- Bishop R F, Davidson G P, Holmes I H, Ruck B J. Virus particles in epithelial cells of duodenal mucosa from children with acute non-bacterial gastroenteritis. Lancet. 1973;2:1281–1283. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

