When are class I metabotropic glutamate receptors necessary for long-term potentiation?
- PMID: 9698302
- PMCID: PMC6793202
- DOI: 10.1523/JNEUROSCI.18-16-06071.1998
When are class I metabotropic glutamate receptors necessary for long-term potentiation?
Abstract
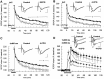
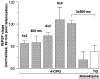
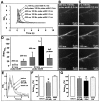
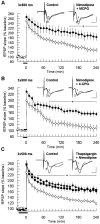
The involvement of metabotropic glutamate receptors (mGluRs) in hippocampal long-term potentiation (LTP) is a matter of controversial debate. Using [Ca2+]i measurements by confocal laser scanning microscopy and field recordings of EPSPs (fEPSPs) in the hippocampal CA1-region, we found that the efficacy of the broad-spectrum mGluR-antagonist (S)-alpha-methyl-4-carboxyphenylglycine (MCPG) and of (S)-4-carboxy-phenylglycine (4-CPG), a selective antagonist at class I mGluRs, in LTP is contingent on the tetanization strength and the resulting [Ca2+]i response. As indicated by experiments in which we blocked voltage-dependent calcium channels (VDCCs) and intracellular Ca2+ stores (ICSs), the functional significance of class I mGluRs in LTP is confined to certain types of potentiation, which are induced by weak tetanization protocols and require the release of Ca2+ from ICSs for induction. During strong tetanic stimulation, this Ca2+ source is functionally bypassed by activating VDCCs.
Figures

References
-
- Aiba A, Chen C, Herrup K, Rosenmund C, Stevens CF, Tonegawa S. Reduced hippocampal long-term potentiation and context-specific deficit in associative learning in mGluR1 mutant mice. Cell. 1994;79:365–375. - PubMed
-
- Artola A, Singer W. Long-term depression of excitatory synaptic transmission and its relationship to long-term potentiation. Trends Neurosci. 1993;16:480–487. - PubMed
-
- Bashir ZI, Bortolotto ZA, Davies CH, Beretta N, Irving AJ, Sea AJ, Henley JM, Jane DE, Watkins JC, Collingridge GL. Induction of LTP in the hippocampus needs synaptic activation of glutamate metabotropic receptors. Nature. 1993;363:347–350. - PubMed
-
- Behnisch T, Reymann KG. Thapsigargin blocks long-term potentiation induced by weak, but not strong tetanization in rat hippocampal CA1 neurons. Neurosci Lett. 1995;192:185–188. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
