Survival of cultured neurons from amyloid precursor protein knock-out mice against Alzheimer's amyloid-beta toxicity and oxidative stress
- PMID: 9698314
- PMCID: PMC6793217
- DOI: 10.1523/JNEUROSCI.18-16-06207.1998
Survival of cultured neurons from amyloid precursor protein knock-out mice against Alzheimer's amyloid-beta toxicity and oxidative stress
Abstract
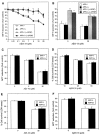
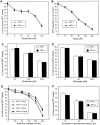
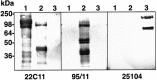
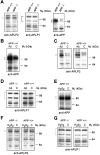
Studies on the amyloid precursor protein (APP) have suggested that it may be neuroprotective against amyloid-beta (Abeta) toxicity and oxidative stress. However, these findings have been obtained from either transfection of cell lines and mice that overexpress human APP isoforms or pretreatment of APP-expressing primary neurons with exogenous soluble APP. The neuroprotective role of endogenously expressed APP in neurons exposed to Abeta or oxidative stress has not been determined. This was investigated using primary cortical and cerebellar neuronal cultures established from APP knock-out (APP-/-) and wild-type (APP+/+) mice. Differences in susceptibility to Abeta toxicity or oxidative stress were not found between APP-/- and APP+/+ neurons. This observation may reflect the expression of the amyloid precursor-like proteins 1 and 2 (APLP1 and APLP2) molecules and supports the theory that APP and the APLPs may have similar functional activities. Increased expression of cell-associated APLP2, but not APLP1, was detected in Abeta-treated APP-/- and APP+/+ cultures but not in H2O2-treated cultures. This suggests that the Abeta toxicity pathway differs from other general forms of oxidative stress. These findings show that Abeta toxicity does not require an interaction of the Abeta peptide with the parental molecule (APP) and is therefore distinct from prion protein neurotoxicity that is dependent on the expression of the parental cellular prion protein.
Figures
Similar articles
-
The Alzheimer's disease amyloid precursor protein modulates copper-induced toxicity and oxidative stress in primary neuronal cultures.J Neurosci. 1999 Nov 1;19(21):9170-9. doi: 10.1523/JNEUROSCI.19-21-09170.1999. J Neurosci. 1999. PMID: 10531420 Free PMC article.
-
Diverse fibrillar peptides directly bind the Alzheimer's amyloid precursor protein and amyloid precursor-like protein 2 resulting in cellular accumulation.Brain Res. 2003 Mar 21;966(2):231-44. doi: 10.1016/s0006-8993(02)04173-2. Brain Res. 2003. PMID: 12618346
-
Mutations in amyloid precursor protein and presenilin-1 genes increase the basal oxidative stress in murine neuronal cells and lead to increased sensitivity to oxidative stress mediated by amyloid beta-peptide (1-42), HO and kainic acid: implications for Alzheimer's disease.J Neurochem. 2006 Mar;96(5):1322-35. doi: 10.1111/j.1471-4159.2005.03647.x. J Neurochem. 2006. PMID: 16478525
-
Amyloid beta-peptide (1-42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer's disease brain. A review.Free Radic Res. 2002 Dec;36(12):1307-13. doi: 10.1080/1071576021000049890. Free Radic Res. 2002. PMID: 12607822 Review.
-
Processing of amyloid precursor protein and amyloid peptide neurotoxicity.Curr Alzheimer Res. 2008 Apr;5(2):92-9. doi: 10.2174/156720508783954721. Curr Alzheimer Res. 2008. PMID: 18393795 Review.
Cited by
-
Oxidative stress, cell cycle, and neurodegeneration.J Clin Invest. 2003 Mar;111(6):785-93. doi: 10.1172/JCI18182. J Clin Invest. 2003. PMID: 12639981 Free PMC article. Review. No abstract available.
-
The Alzheimer's disease amyloid precursor protein modulates copper-induced toxicity and oxidative stress in primary neuronal cultures.J Neurosci. 1999 Nov 1;19(21):9170-9. doi: 10.1523/JNEUROSCI.19-21-09170.1999. J Neurosci. 1999. PMID: 10531420 Free PMC article.
-
Inclusion body myositis: a view from the Caenorhabditis elegans muscle.Mol Neurobiol. 2008 Oct;38(2):178-98. doi: 10.1007/s12035-008-8041-0. Epub 2008 Sep 5. Mol Neurobiol. 2008. PMID: 18773311 Review.
-
The blockage of the Nogo/NgR signal pathway in microglia alleviates the formation of Aβ plaques and tau phosphorylation in APP/PS1 transgenic mice.J Neuroinflammation. 2016 Mar 3;13(1):56. doi: 10.1186/s12974-016-0522-x. J Neuroinflammation. 2016. PMID: 26939570 Free PMC article.
-
Suppression of cyclin-dependent kinase 5 activation by amyloid precursor protein: a novel excitoprotective mechanism involving modulation of tau phosphorylation.J Neurosci. 2005 Dec 14;25(50):11542-52. doi: 10.1523/JNEUROSCI.3831-05.2005. J Neurosci. 2005. PMID: 16354912 Free PMC article.
References
-
- Abe K, Kimura H. Amyloid β toxicity consists of a Ca2+-independent early phase and a Ca2+-dependent late phase. J Neurochem. 1996;67:2074–2078. - PubMed
-
- Barger SW, Fiscus RR, Ruth P, Hofmann F, Mattson MP. Role of cyclic GMP in the regulation of neuronal calcium and survival by secreted forms of β-amyloid precursor. J Neurochem. 1995;64:2087–2096. - PubMed
-
- Brown DR, Schmidt B, Kretzschmar HA. A neurotoxic prion protein fragment enhances proliferation of microglia but not astrocytes in culture. Glia. 1996a;18:59–67. - PubMed
-
- Brown DR, Schmidt B, Kretzschmar HA. Role of microglia and host prion protein in neurotoxicity of a prion protein fragment. Nature. 1996b;380:345–347. - PubMed
-
- Brunden KR, Richter-Cook NJ, Chaturvedi N, Frederickson RCA. pH-dependent binding of synthetic β-amyloid peptides to glycosaminoglycans. J Neurochem. 1993;61:2147–2154. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
