Neuronal correlates of amblyopia in the visual cortex of macaque monkeys with experimental strabismus and anisometropia
- PMID: 9698332
- PMCID: PMC6793177
- DOI: 10.1523/JNEUROSCI.18-16-06411.1998
Neuronal correlates of amblyopia in the visual cortex of macaque monkeys with experimental strabismus and anisometropia
Abstract
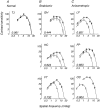
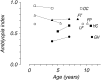
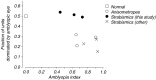
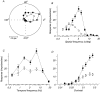
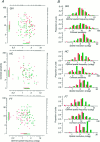
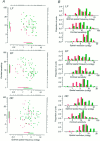
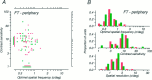
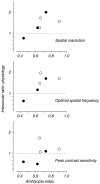
Amblyopia is a developmental disorder of pattern vision. After surgical creation of esotropic strabismus in the first weeks of life or after wearing -10 diopter contact lenses in one eye to simulate anisometropia during the first months of life, macaques often develop amblyopia. We studied the response properties of visual cortex neurons in six amblyopic macaques; three monkeys were anisometropic, and three were strabismic. In all monkeys, cortical binocularity was reduced. In anisometropes, the amblyopic eye influenced a relatively small proportion of cortical neurons; in strabismics, the influence of the two eyes was more nearly equal. The severity of amblyopia was related to the relative strength of the input of the amblyopic eye to the cortex only for the more seriously affected amblyopes. Measurements of the spatial frequency tuning and contrast sensitivity of cortical neurons showed few differences between the eyes for the three less severe amblyopes (two strabismic and one anisometropic). In the three more severely affected animals (one strabismic and two anisometropic), the optimal spatial frequency and spatial resolution of cortical neurons driven by the amblyopic eye were substantially and significantly lower than for neurons driven by the nonamblyopic eye. There were no reliable differences in neuronal contrast sensitivity between the eyes. A sample of neurons recorded from cortex representing the peripheral visual field showed no interocular differences, suggesting that the effects of amblyopia were more pronounced in portions of the cortex subserving foveal vision. Qualitatively, abnormalities in both the eye dominance and spatial properties of visual cortex neurons were related on a case-by-case basis to the depth of amblyopia. Quantitative analysis suggests, however, that these abnormalities alone do not explain the full range of visual deficits in amblyopia. Studies of extrastriate cortical areas may uncover further abnormalities that explain these deficits.
Figures


References
-
- Baker FH, Grigg P, von Noorden GK. Effects of visual deprivation and strabismus on the response of neurons in the visual cortex of the monkey, including studies of striate and prestriate cortex in the normal animal. Brain Res. 1974;66:185–208.
-
- Boothe RG, Kiorpes L, Williams RA, Teller DY. Operant measurements of contrast sensitivity in infant macaque monkeys during normal development. Vision Res. 1989;28:387–396. - PubMed
-
- Crawford ML, Harwerth RS, Chino YM, Smith EL. Binocularity in prism-reared monkeys. Eye. 1996;10:161–166. - PubMed
-
- Crawford MLJ, von Noorden GK. The effects of short-term experimental strabismus on the visual system in Macaca mulatta. Invest Ophthalmol Vis Sci. 1979;18:496–505. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
