Detection of a novel strain of porcine circovirus in pigs with postweaning multisystemic wasting syndrome
- PMID: 9705388
- PMCID: PMC105158
- DOI: 10.1128/JCM.36.9.2535-2541.1998
Detection of a novel strain of porcine circovirus in pigs with postweaning multisystemic wasting syndrome
Abstract
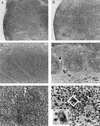
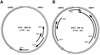
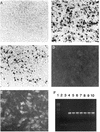
Swine infectious agents, especially viruses, are potential public health risks associated with the use of pig organs for xenotransplantation in humans. Therefore, there is a need for better characterization of swine viruses and for the development of diagnostic tests for their detection. We report here isolation of a novel strain of porcine circovirus (PCV) from pigs with postweaning multisystemic wasting syndrome (PMWS). Affected pigs exhibited severe interstitial pneumonia and lymphoid depletion. The complete nucleotide sequence (1,768 nucleotides) of the genome of the PCV isolate was determined and compared with the sequence of the PCV strain isolated from PK-15 cells. Sequence comparison revealed significant differences between the two PCV strains, with an overall DNA homology of 76%. Two major open reading frames (ORFs) were identified. ORF1 was more conserved between the two strains, with 83% nucleotide homology and 86% amino acid homology. ORF2 was more variable, with nucleotide homology of 67% and amino acid homology of 65%. PCR and in situ hybridization demonstrated abundant viral DNA in various organs of pigs with PMWS. In situ hybridization demonstrated that this strain of PCV targets multiple organs and infects macrophages, lymphocytes, endothelial cells, and epithelial cells.
Figures

References
-
- Albina E. Epidemiology of porcine reproductive and respiratory syndrome (PRRS): an overview. Vet Microbiol. 1997;55:309–316. - PubMed
-
- Allan G M, McNeilly F, Cassidy J P, Reilly G A C, Adair B, Ellis W A, McNulty M S. Pathogenesis of porcine circovirus, experimental infections of colostrum deprived piglets and examination of pig fetal material. Vet Microbiol. 1995;44:49–64. - PubMed
-
- Allan G M, Phenix K V, Todd D, McNulty M S. Some biological and physico-chemical properties of Porcine Circovirus. J Vet Med B. 1994;41:17–26. - PubMed
-
- Boevink P, Chu P W G, Keese P. Sequence of Subterranean Clover Stunt Virus DNA: affinities with the Geminiviruses. Virology. 1995;207:354–361. - PubMed
-
- Botner A. Diagnosis of PRRS. Vet Microbiol. 1997;55:295–301. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

