Cloning and characterization of multigenes encoding the immunodominant 30-kilodalton major outer membrane proteins of Ehrlichia canis and application of the recombinant protein for serodiagnosis
- PMID: 9705412
- PMCID: PMC105182
- DOI: 10.1128/JCM.36.9.2671-2680.1998
Cloning and characterization of multigenes encoding the immunodominant 30-kilodalton major outer membrane proteins of Ehrlichia canis and application of the recombinant protein for serodiagnosis
Abstract
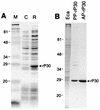
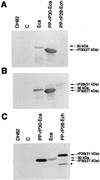
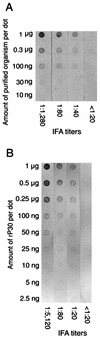
A 30-kDa major outer membrane protein of Ehrlichia canis, the agent of canine ehrlichiosis, is the major antigen recognized by both naturally and experimentally infected dog sera. The protein cross-reacts with a serum against a recombinant 28-kDa protein (rP28), one of the outer membrane proteins of a gene (omp-1) family of Ehrlichia chaffeensis. Two DNA fragments of E. canis were amplified by PCR with two primer pairs based on the sequences of E. chaffeensis omp-1 genes, cloned, and sequenced. Each fragment contained a partial 30-kDa protein gene of E. canis. Genomic Southern blot analysis with the partial gene probes revealed the presence of multiple copies of these genes in the E. canis genome. Three copies of the entire gene (p30, p30-1, and p30a) were cloned and sequenced from the E. canis genomic DNA. The open reading frames of the two copies (p30 and p30-1) were tandemly arranged with an intergenic space. The three copies were similar but not identical and contained a semivariable region and three hypervariable regions in the protein molecules. The following genes homologous to three E. canis 30-kDa protein genes and the E. chaffeensis omp-1 family were identified in the closely related rickettsiae: wsp from Wolbachia sp. , p44 from the agent of human granulocytic ehrlichiosis, msp-2 and msp-4 from Anaplasma marginale, and map-1 from Cowdria ruminantium. Phylogenetic analysis among the three E. canis 30-kDa proteins and the major surface proteins of the rickettsiae revealed that these proteins are divided into four clusters and the two E. canis 30-kDa proteins are closely related but that the third 30-kDa protein is not. The p30 gene was expressed as a fusion protein, and the antibody to the recombinant protein (rP30) was raised in a mouse. The antibody reacted with rP30 and a 30-kDa protein of purified E. canis. Twenty-nine indirect fluorescent antibody (IFA)-positive dog plasma specimens strongly recognized the rP30 of E. canis. To evaluate whether the rP30 is a suitable antigen for serodiagnosis of canine ehrlichiosis, the immunoreactions between rP30 and the whole purified E. canis antigen were compared in the dot immunoblot assay. Dot reactions of both antigens with IFA-positive dog plasma specimens were clearly distinguishable by the naked eye from those with IFA-negative plasma specimens. By densitometry with a total of 42 IFA-positive and -negative plasma specimens, both antigens produced results similar in sensitivity and specificity. These findings suggest that the rP30 antigen provides a simple, consistent, and rapid serodiagnosis for canine ehrlichiosis. Cloning of multigenes encoding the 30-kDa major outer membrane proteins of E. canis will greatly facilitate understanding pathogenesis and immunologic study of canine ehrlichosis and provide a useful tool for phylogenetic analysis.
Figures






References
-
- Buhles W C, Huxsoll D L, Ristic M. Tropical canine pancytopenia: clinical, haematologic, and serologic, and serologic response of dogs to Ehrlichia canis infection, tetracycline therapy, and challenge inoculation. J Infect Dis. 1974;130:358–367. - PubMed
-
- Cadman H F, Kelly P J, Matthewman L A, Zhou R, Mason P R. Comparison of the dot-blot enzyme linked immunoassay with immunofluorescence for detecting antibodies to Ehrlichia canis. Vet Rec. 1994;135:362. - PubMed
-
- Codner E C, Farris-Smith L L. Characterization of the subclinical phase of ehrlichiosis in dogs. J Am Vet Med Assoc. 1986;189:47–50. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

