Ca2+ influx through voltage-gated Ca2+ channels regulates 5-HT3 receptor channel desensitization in rat glioma x mouse neuroblastoma hybrid NG108-15 cells
- PMID: 9705989
- PMCID: PMC2231060
- DOI: 10.1111/j.1469-7793.1998.361bk.x
Ca2+ influx through voltage-gated Ca2+ channels regulates 5-HT3 receptor channel desensitization in rat glioma x mouse neuroblastoma hybrid NG108-15 cells
Abstract
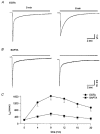
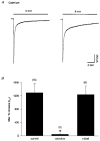
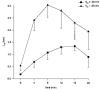
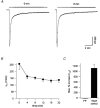
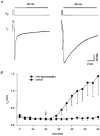
1. The kinetics of desensitization of the 5-HT3 receptor (5-HT3R)-gated ion channel were investigated using whole-cell and perforated-patch recording techniques in NG108-15 cells. 2. Rapid application of 5-HT (50 microM) elicited a 5-HT3R-mediated inward current response that desensitized completely in the continued presence of agonist. In the whole-cell recording configuration (holding potential of -70 mV) while buffering internal calcium (Cai2+) with 5 mM EGTA (0.5 mM added Ca2+; with an estimated free [Ca2+] of 30 nM), the rate of desensitization was initially rapid (with a half-time of approximately 230 ms), but dramatically slowed with time by 1120 +/- 160%. 3. This slowing in the rate of desensitization was reduced by stronger Ca2+ buffering (20 mM BAPTA, without added Ca2+), or by the bath application of cadmium (100 microM) to block voltage-gated Ca2+ channels. The rate of desensitization was also dependent on membrane potential. 4. In perforated-patch recordings, the rate of desensitization remained constant. However, a slowing in the desensitization rate could be induced by depolarizing cells immediately prior to the application of 5-HT. 5. The depolarization-induced slowing was blocked by incubating cells with BAPTA-AM (a membrane-permeant analogue of BAPTA) or by the bath application of cadmium. 6. These data suggest that Ca2+ influx through a cadmium-sensitive voltage-gated Ca2+ channel increases the cytoplasmic Ca2+ concentration ([Ca2+]i) and induces a dramatic slowing in the kinetics of desensitization of the 5-HT3R channel. These data provide evidence for cross-talk between voltage-gated Ca2+ channels and 5-HT3Rs in NG108-15 cells.
Figures

Similar articles
-
A study of the voltage dependence of capsaicin-activated membrane currents in rat sensory neurones before and after acute desensitization.J Physiol. 1999 Aug 1;518 ( Pt 3)(Pt 3):721-33. doi: 10.1111/j.1469-7793.1999.0721p.x. J Physiol. 1999. PMID: 10420009 Free PMC article.
-
High calcium permeability of serotonin 5-HT3 receptors on presynaptic nerve terminals from rat striatum.J Neurochem. 1998 Mar;70(3):1094-103. doi: 10.1046/j.1471-4159.1998.70031094.x. J Neurochem. 1998. PMID: 9489730
-
Calcium influx through voltage-gated calcium channels regulates 5-HT3 receptor channel desensitization in NG108-15 cells.Ann N Y Acad Sci. 1998 Dec 15;861:253-4. doi: 10.1111/j.1749-6632.1998.tb10208.x. Ann N Y Acad Sci. 1998. PMID: 9928274 No abstract available.
-
Calcium channels: unanswered questions.J Bioenerg Biomembr. 2003 Dec;35(6):461-75. doi: 10.1023/b:jobb.0000008020.86004.28. J Bioenerg Biomembr. 2003. PMID: 15000516 Review.
-
Effects of toxic environmental contaminants on voltage-gated calcium channel function: from past to present.J Bioenerg Biomembr. 2003 Dec;35(6):507-32. doi: 10.1023/b:jobb.0000008023.11211.13. J Bioenerg Biomembr. 2003. PMID: 15000519 Review.
Cited by
-
Characteristics for enhanced response of serotonin-evoked ion dynamics in differentiated NG108-15 cells.Neurochem Res. 2009 May;34(5):1011-9. doi: 10.1007/s11064-008-9839-x. Epub 2008 Dec 12. Neurochem Res. 2009. PMID: 19082885
-
Length and amino acid sequence of peptides substituted for the 5-HT3A receptor M3M4 loop may affect channel expression and desensitization.PLoS One. 2012;7(4):e35563. doi: 10.1371/journal.pone.0035563. Epub 2012 Apr 23. PLoS One. 2012. PMID: 22539982 Free PMC article.
-
Open probability of homomeric murine 5-HT3A serotonin receptors depends on subunit occupancy.J Physiol. 2001 Sep 1;535(Pt 2):427-43. doi: 10.1111/j.1469-7793.2001.00427.x. J Physiol. 2001. PMID: 11533135 Free PMC article.
-
Serotonin 5-HT3 receptor-mediated vomiting occurs via the activation of Ca2+/CaMKII-dependent ERK1/2 signaling in the least shrew (Cryptotis parva).PLoS One. 2014 Aug 14;9(8):e104718. doi: 10.1371/journal.pone.0104718. eCollection 2014. PLoS One. 2014. PMID: 25121483 Free PMC article.
-
Serotonin 5-HT(3) receptors in rat CA1 hippocampal interneurons: functional and molecular characterization.J Physiol. 2002 Nov 1;544(3):715-26. doi: 10.1113/jphysiol.2002.029736. J Physiol. 2002. PMID: 12411518 Free PMC article.
References
-
- Brown DA, Docherty RJ, McFadzean I. Calcium channels in vertebrate neurons. Experiments on a neuroblastoma hybrid model. Annals of the New York Academy of Sciences. 1989;560:358–372. - PubMed
-
- Caulfield MP, Robbins J, Brown DA. Neurotransmitters inhibit the omega-conotoxin-sensitive component of Ca current in neuroblastoma × glioma hybrid (NG 108–15) cells, not the nifedipine-sensitive component. Pflügers Archiv. 1992;420:486–492. - PubMed
-
- Chueh S-H, Kao L-S. Extracellular ATP stimulates calcium influx in neuroblastoma × glioma hybrid NG108-15 cells. Journal of Neurochemistry. 1993;61:1782–1788. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

