The striatal neurotensin receptor modulates striatal and pallidal glutamate and GABA release: functional evidence for a pallidal glutamate-GABA interaction via the pallidal-subthalamic nucleus loop
- PMID: 9712666
- PMCID: PMC6792956
- DOI: 10.1523/JNEUROSCI.18-17-06977.1998
The striatal neurotensin receptor modulates striatal and pallidal glutamate and GABA release: functional evidence for a pallidal glutamate-GABA interaction via the pallidal-subthalamic nucleus loop
Abstract
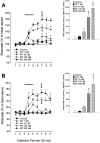
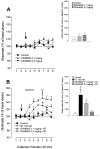
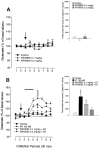
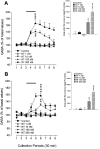
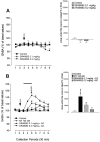
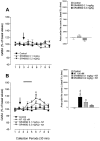
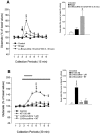
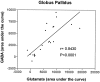
In the present study, we used dual-probe microdialysis to investigate the effects of intrastriatal perfusion with neurotensin (NT) on striatal and pallidal glutamate and GABA release. The role of the pallidal GABAA receptor in the intrastriatal NT-induced increase in pallidal glutamate release was also investigated. Intrastriatal NT (100 and 300 nM) increased striatal glutamate and GABA (100 nM, 155 +/- 9 and 141 +/- 6%, respectively; 300 nM, 179 +/- 8 and 166 +/- 11%, respectively) release, as well as pallidal glutamate and GABA (100 nM, 144 +/- 8 and 130 +/- 5%; 300 nM, 169 +/- 9 and 157 +/- 8%, respectively) release. These effects were dose-dependently antagonized by the NT receptor antagonist 2-[(1-(7-chloro-4-quinolinyl)-5-(2, 6-dimethoxy-phenyl)pyrazol-3-yl)carboxylamino]tricyclo)3.3.1 .1.3. 7)-decan-2-carboxylic acid (SR48692). Intrasubthalamic injection of the GABAA receptor antagonist (-)-bicuculline (10 pmol/100 nl, 30 sec) rapidly increased pallidal glutamate release, whereas the intrastriatal NT-induced increase in pallidal glutamate release was counteracted by intrapallidal perfusion with (-)-bicuculline, suggesting that an increase in striopallidal GABA-mediated inhibition of the GABAergic pallidal-subthalamic pathway results in an increased glutamatergic drive in the subthalamic-pallidal pathway. These results demonstrate a tonic pallidal GABA-mediated inhibition of excitatory subthalamic-pallidal neurons and strengthen the evidence for a functional role of NT in the regulation of glutamate and GABA transmission in the basal ganglia. The ability of intrastriatal SR48692 to counteract the NT-induced increase in both striatal and pallidal glutamate and GABA release suggests that blockade of the striatal NT receptor may represent a possible new therapeutic strategy in the treatment of those hypokinetic disorders implicated in disorders of the indirect pathway mediating motor inhibition.
Figures

References
-
- Agnati LF, Fuxe K, Benfenati F, Battistini N. Neurotensin in vitro markedly reduces the affinity in subcortical limbic [3H]N-propylnorapomorphine binding sites. Acta Physiol Scand. 1983;117:299–301. - PubMed
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Alexander GE, Crutcher M. Functional architecture of basal ganglia circuits: neuronal substrates of parallel processing. Trends Neurosci. 1990;13:266–271. - PubMed
-
- Amalric M, Farin D, Dormont JF, Schmied A. GABA-receptor activation in the globus pallidus and entopeduncolar nucleus: opposite effects on reaction time performance in the cat. Exp Brain Res. 1994;102:244–258. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
