Plasma hydroxy metronidazole/metronidazole ratio in anti-HCV carriers with and without apparent liver disease
- PMID: 9723829
- PMCID: PMC1873660
- DOI: 10.1046/j.1365-2125.1998.00758.x
Plasma hydroxy metronidazole/metronidazole ratio in anti-HCV carriers with and without apparent liver disease
Abstract
Aims: To evaluate plasma hydroxy-metronidazole/metronidazole ratio as a dynamic liver function test in HCV-infected individuals with/without liver disease, in the absence of liver cirrhosis.
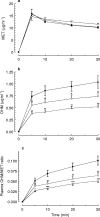
Methods: Metronidazole was administered intravenously in healthy volunteers, asymptomatic anti-HCV-positive blood donors, and in chronic hepatitis C patients. Serology to HCV was determined by a second generation assay and confirmed by gelatin particle agglutination test using recombinant antigens C22-3 and C200. Plasma concentration of metronidazole and hydroxy-metronidazole was measured by high performance liquid chromatography in samples collected 5, 10, 20 and 30 min following the end of metronidazole infusion.
Results: Chronic hepatitis C patients had abnormal liver enzymes, while healthy volunteers and anti-HCV-positive blood donors had normal liver biochemistry tests. Plasma metronidazole concentration was similar in all groups studied. Plasma hydroxy-metronidazole/metronidazole ratio was significantly reduced in HCV-infected subjects, an effect observed 10 min after the end of drug infusion.
Conclusions: Metronidazole clearance is impaired in anti-HCV-positive blood donors and chronic hepatitis C patients, indicating that HCV is capable of affecting liver function at early stages of the disease. The metronidazole clearance test can detect impaired liver function in HCV-infected individuals even in the absence of liver cirrhosis.
Figures
Similar articles
-
Plasma hydroxy-metronidazole/metronidazole ratio in hepatitis C virus-induced liver disease.Braz J Med Biol Res. 2005 Mar;38(3):437-44. doi: 10.1590/s0100-879x2005000300015. Epub 2005 Mar 8. Braz J Med Biol Res. 2005. PMID: 15761624
-
Plasma hydroxy-metronidazole/metronidazole ratio can detect early changes in hepatic function in ethanol-induced liver injury.Aliment Pharmacol Ther. 1999 Oct;13(10):1335-41. doi: 10.1046/j.1365-2036.1999.00620.x. Aliment Pharmacol Ther. 1999. PMID: 10540049
-
Plasma hydroxy-metronidazole/metronidazole ratio in patients with liver disease and in healthy volunteers.Br J Clin Pharmacol. 1995 Nov;40(5):477-80. doi: 10.1111/j.1365-2125.1995.tb05792.x. Br J Clin Pharmacol. 1995. PMID: 8703652 Free PMC article. Clinical Trial.
-
Clinical importance of assessment of anti-HCV IgM antibodies in chronic hepatitis C.Acta Virol. 1996 Apr;40(2):61-5. Acta Virol. 1996. PMID: 8886113
-
Routes of infection, viremia, and liver disease in blood donors found to have hepatitis C virus infection.N Engl J Med. 1996 Jun 27;334(26):1691-6. doi: 10.1056/NEJM199606273342602. N Engl J Med. 1996. PMID: 8637513
References
-
- Kuo G, Choo QL, Alter HJ, et al. An assay for circulating antibodies to a major etiologic virus of human non-A, non-B hepatitis. Science. 1989;244:359–362. - PubMed
-
- Dusheiko GM. Hepatitis C virus. In: Pounder RE, editor. Recent advances in gastroenterology. 9. Edinburgh: Churchill Livingstone; 1992. pp. 195–216.
-
- Alter MJ, Margolis HS, Krawczynsky K, et al. The natural history of community-acquired hepatitis C in the United States. New Engl J Med. 1992;327:1899–1905. - PubMed
-
- Alberti A, Morsica G, Chemello L, et al. Hepatitis C viraemia and liver disease in symptom-free individuals with anti HCV. Lancet. 1992;340:697–698. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

