Altering the anaerobic transcription factor FNR confers a hemolytic phenotype on Escherichia coli K12
- PMID: 9724723
- PMCID: PMC27914
- DOI: 10.1073/pnas.95.18.10449
Altering the anaerobic transcription factor FNR confers a hemolytic phenotype on Escherichia coli K12
Abstract
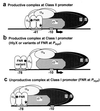
The recent outbreaks of Escherichia coli O157-associated food poisoning have focused attention on the virulence determinants of E. coli. Here, it is reported that single base substitutions in the fnr gene encoding the oxygen-responsive transcription regulator FNR (fumarate and nitrate reduction regulator) are sufficient to confer a hemolytic phenotype on E. coli K12, the widely used laboratory strain. The mechanism involves enhancing the expression of a normally dormant hemolysin gene (hlyE) located in the E. coli chromosome. The mutations direct single amino acid substitutions in the activating regions (AR1 and AR3) of FNR that contact RNA polymerase. It is concluded that altering a resident transcription regulator, or acquisition of a competent heterologous regulator, could generate a pool of hemolytic, and therefore more virulent, strains of E. coli in nature.
Figures

Similar articles
-
The molecular basis for the differential regulation of the hlyE-encoded haemolysin of Escherichia coli by FNR and HlyX lies in the improved activating region 1 contact of HlyX.Microbiology (Reading). 1997 Dec;143 ( Pt 12):3785-3793. doi: 10.1099/00221287-143-12-3785. Microbiology (Reading). 1997. PMID: 9421903
-
HlyX, the FNR homologue of Actinobacillus pleuropneumoniae, is a [4Fe-4S]-containing oxygen-responsive transcription regulator that anaerobically activates FNR-dependent class I promoters via an enhanced AR1 contact.Mol Microbiol. 1997 May;24(3):593-605. doi: 10.1046/j.1365-2958.1997.3801737.x. Mol Microbiol. 1997. PMID: 9179852
-
Downregulation of Escherichia coli yfiD expression by FNR occupying a site at -93.5 involves the AR1-containing face of FNR.Mol Microbiol. 1998 Aug;29(4):1113-23. doi: 10.1046/j.1365-2958.1998.01002.x. Mol Microbiol. 1998. PMID: 9767578
-
FNR and its role in oxygen-regulated gene expression in Escherichia coli.FEMS Microbiol Rev. 1990 Aug;6(4):399-428. doi: 10.1111/j.1574-6968.1990.tb04109.x. FEMS Microbiol Rev. 1990. PMID: 2248796 Review.
-
Oxygen regulated gene expression in Escherichia coli: control of anaerobic respiration by the FNR protein.Antonie Van Leeuwenhoek. 1991 Feb;59(2):65-76. doi: 10.1007/BF00445650. Antonie Van Leeuwenhoek. 1991. PMID: 1854188 Review.
Cited by
-
Transcription activation by FNR: evidence for a functional activating region 2.J Bacteriol. 2002 Nov;184(21):5855-61. doi: 10.1128/JB.184.21.5855-5861.2002. J Bacteriol. 2002. PMID: 12374818 Free PMC article.
-
Alternate SlyA and H-NS nucleoprotein complexes control hlyE expression in Escherichia coli K-12.Mol Microbiol. 2007 Nov;66(3):685-98. doi: 10.1111/j.1365-2958.2007.05950.x. Epub 2007 Sep 24. Mol Microbiol. 2007. PMID: 17892462 Free PMC article.
-
Optimization of overexpression of a chaperone protein of steroid C25 dehydrogenase for biochemical and biophysical characterization.Protein Expr Purif. 2017 Jun;134:47-62. doi: 10.1016/j.pep.2017.03.019. Epub 2017 Mar 23. Protein Expr Purif. 2017. PMID: 28343996 Free PMC article.
-
Cytolysin gene expression in Enterococcus faecalis is regulated in response to aerobiosis conditions.Mol Genet Genomics. 2003 Apr;269(1):31-9. doi: 10.1007/s00438-003-0819-1. Epub 2003 Feb 22. Mol Genet Genomics. 2003. PMID: 12715151
-
Novel denitrifying bacterium Ochrobactrum anthropi YD50.2 tolerates high levels of reactive nitrogen oxides.Appl Environ Microbiol. 2009 Aug;75(16):5186-94. doi: 10.1128/AEM.00604-09. Epub 2009 Jun 19. Appl Environ Microbiol. 2009. PMID: 19542343 Free PMC article.
References
-
- Guest J R, Green J, Irvine A S, Spiro S. In: Regulation of Gene Expression in Escherichia coli. Lin E C C, Lynch A S, editors. Austin, TX: Landes; 1996. pp. 317–342.
-
- Lazazzera B A, Beinert H, Khoroshilova N, Kennedy M C, Kiley P J. J Biol Chem. 1996;271:2762–2768. - PubMed
-
- Jordan P, Thomson A J, Ralph E T, Guest J R, Green J. FEBS Lett. 1997;416:349–352. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

