In vitro activity of lumefantrine (benflumetol) against clinical isolates of Plasmodium falciparum in Yaoundé, Cameroon
- PMID: 9736561
- PMCID: PMC105831
- DOI: 10.1128/AAC.42.9.2347
In vitro activity of lumefantrine (benflumetol) against clinical isolates of Plasmodium falciparum in Yaoundé, Cameroon
Abstract
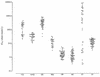
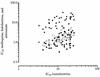
The in vitro antimalarial activity of the new Chinese synthetic drug, lumefantrine, also known as benflumetol (a fluorene derivative belonging to the aminoalcohol class), was determined by an isotopic microtest against 61 fresh clinical isolates of Plasmodium falciparum and compared with that of other established antimalarial agents. The geometric mean 50% inhibitory concentration of lumefantrine was 11.9 nmol/liter (95% confidence intervals, 10.4 to 13.6 nmol/liter; range, 3.3 to 25.6 nmol/liter). The in vitro activities of lumefantrine against the chloroquine-sensitive and the chloroquine-resistant isolates did not differ (P > 0.05). There was a significant positive correlation of responses between lumefantrine and two other aminoalcohols studied, mefloquine (r = 0.688) and halofantrine (r = 0.677), and between lumefantrine and artesunate (r = 0.420), suggesting a potential for in vitro cross-resistance. Our data suggest high in vitro activity of lumefantrine, comparable to that of mefloquine, and are in agreement with the promising results of preliminary clinical trials.
Figures

References
-
- Basco L K, Le Bras J. In vitro activity of halofantrine and its relationship to other standard antimalarial drugs against African isolates and clones of Plasmodium falciparum. Am J Trop Med Hyg. 1992;47:521–527. - PubMed
-
- Basco L K, Le Bras J. In vitro activity of pyronaridine against African strains of Plasmodium falciparum. Ann Trop Med Parasitol. 1992;86:447–454. - PubMed
-
- Basco L K, Le Bras J. In vitro activities of monodesethylamodiaquine and amopyroquine against African isolates and clones of Plasmodium falciparum. Am J Trop Med Hyg. 1993;48:120–125. - PubMed
-
- Basco L K, Le Bras J. In vitro activity of artemisinin derivatives against African isolates and clones of Plasmodium falciparum. Am J Trop Med Hyg. 1993;49:301–307. - PubMed
-
- Basco L K, Ramiliarisoa O, Le Bras J. In vitro activity of pyrimethamine, cycloguanil, and other antimalarial drugs against African isolates and clones of Plasmodium falciparum. Am J Trop Med Hyg. 1994;50:193–199. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

