Safety, pharmacokinetics, and antiretroviral activity of intravenous 9-[2-(R)-(Phosphonomethoxy)propyl]adenine, a novel anti-human immunodeficiency virus (HIV) therapy, in HIV-infected adults
- PMID: 9736567
- PMCID: PMC105837
- DOI: 10.1128/AAC.42.9.2380
Safety, pharmacokinetics, and antiretroviral activity of intravenous 9-[2-(R)-(Phosphonomethoxy)propyl]adenine, a novel anti-human immunodeficiency virus (HIV) therapy, in HIV-infected adults
Abstract
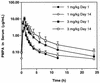
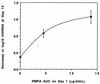
9-[2-(R)-(Phosphonomethoxy)propyl]adenine (PMPA) is a nucleotide analogue with potent antiretroviral activity in vitro and in simian models. A randomized, double-blind, placebo-controlled, dose-escalation clinical trial of intravenous PMPA monotherapy was conducted in 20 human immunodeficiency virus (HIV)-infected adults with CD4 cell counts of >/=200 cells/mm3 and plasma HIV RNA levels of >/=10,000 copies/ml. Two dose levels were evaluated (1 and 3 mg/kg of body weight/day). Ten subjects were enrolled at each dose level (eight randomized to receive PMPA and two randomized to receive placebo). On day 1, a single dose of PMPA or placebo was administered by intravenous infusion. Beginning on study day 8, PMPA or placebo was administered once daily for an additional 7 consecutive days. All subjects tolerated dosing without significant adverse events. Mean peak serum PMPA concentrations were 2.7 +/- 0.9 and 9.1 +/- 2.1 microgram/ml in the 1- and 3-mg/kg cohorts, respectively. Serum concentrations declined in a biexponential fashion, with a terminal half-life of 4 to 8 h. At 3 mg/kg/day, a single infusion of PMPA resulted in a 0.4 log10 median decline in plasma HIV RNA by study day 8. Following 7 consecutive days of study drug administration thereafter, the median changes in plasma HIV RNA from baseline were -1.1, -0.6, and 0.1 log10 in the 3-mg/kg/day, 1-mg/kg/day, and placebo dose groups, respectively. Following the final dose in the 3-mg/kg/day cohort, the reduction in HIV RNA was sustained for 7 days before returning toward baseline. Further studies evaluating an oral prodrug of PMPA are under way.
Figures

Similar articles
-
Administration of 9-[2-(phosphonomethoxy)propyl]adenine (PMPA) for prevention of perinatal simian immunodeficiency virus infection in rhesus macaques.AIDS Res Hum Retroviruses. 1998 Jun 10;14(9):761-73. doi: 10.1089/aid.1998.14.761. AIDS Res Hum Retroviruses. 1998. PMID: 9643376
-
Phase i/ii trial of the pharmacokinetics, safety, and antiretroviral activity of tenofovir disoproxil fumarate in human immunodeficiency virus-infected adults.Antimicrob Agents Chemother. 2001 Oct;45(10):2733-9. doi: 10.1128/AAC.45.10.2733-2739.2001. Antimicrob Agents Chemother. 2001. PMID: 11557462 Free PMC article. Clinical Trial.
-
Pharmacokinetics and bioavailability of the anti-human immunodeficiency virus nucleotide analog 9-[(R)-2-(phosphonomethoxy)propyl]adenine (PMPA) in dogs.Antimicrob Agents Chemother. 1998 Mar;42(3):687-90. doi: 10.1128/AAC.42.3.687. Antimicrob Agents Chemother. 1998. PMID: 9517952 Free PMC article.
-
Tenofovir: a nucleotide analog for the management of human immunodeficiency virus infection.Pharmacotherapy. 2003 Jan;23(1):29-43. doi: 10.1592/phco.23.1.29.31915. Pharmacotherapy. 2003. PMID: 12523458 Review.
-
Tenofovir disoproxil fumarate: a nucleotide reverse transcriptase inhibitor for the treatment of HIV infection.Clin Ther. 2002 Oct;24(10):1515-48. doi: 10.1016/s0149-2918(02)80058-3. Clin Ther. 2002. PMID: 12462284 Review.
Cited by
-
Relative bioavailability study of a novel prodrug of tenofovir, tenofovir dipivoxil fumarate, in healthy male fasted volunteers.Clin Drug Investig. 2012 May 1;32(5):333-8. doi: 10.2165/11599910-000000000-00000. Clin Drug Investig. 2012. PMID: 22413748 Clinical Trial.
-
Nucleotide analogue prodrug tenofovir disoproxil enhances lymphoid cell loading following oral administration in monkeys.Mol Pharm. 2009 Jul-Aug;6(4):1145-51. doi: 10.1021/mp900036s. Mol Pharm. 2009. PMID: 19545170 Free PMC article.
-
From in vitro EC₅₀ to in vivo dose-response for antiretrovirals using an HIV disease model. Part I: a framework.J Pharmacokinet Pharmacodyn. 2012 Aug;39(4):357-68. doi: 10.1007/s10928-012-9255-3. Epub 2012 Jun 27. J Pharmacokinet Pharmacodyn. 2012. PMID: 22736302 Clinical Trial.
-
Efficacy of Tenofovir 1% Vaginal Gel in Reducing the Risk of HIV-1 and HSV-2 Infection.Clin Med Insights Womens Health. 2014 Feb 13;7:1-8. doi: 10.4137/CMWH.S10353. eCollection 2014. Clin Med Insights Womens Health. 2014. PMID: 24741339 Free PMC article. Review.
-
Tackling HIV and AIDS: contributions by non-human primate models.Lab Anim (NY). 2017 May 22;46(6):259-270. doi: 10.1038/laban.1279. Lab Anim (NY). 2017. PMID: 28530684
References
-
- Arimilli M N, Kim C U, Dougherty J, Mulato A, Oliyai R, Shaw J P, Crundy K U, Bischofberger N. Synthesis, in vitro biological evaluation and oral bioavailability of 9-[2-(phosphonomethoxy)propyl]adenine (PMPA) prodrugs. Antivir Chem Chemother. 1997;8:557–564.
-
- Balzarini J, Perno C, Schols D, De Clercq E. Activity of acyclic phosphonate analogues against human immunodeficiency virus in monocytes/macrophages and peripheral blood lymphocytes. Biochem Biophys Res Commun. 1991;178:329–335. - PubMed
-
- Cherrington J M, Chandok R, Mulato A S, Lamy P D, Mitsuya H, Wainberg M. Abstracts of the 37th Interscience Conference on Antimicrobial Agents and Chemotherapy. 1997. In vitro selection and characterization of HIV-1 variants with reduced susceptibility to PMPA, abstr. I-113; p. 264. Toronto, Ontario, Canada.
-
- Danner S A, Carr A, Leonard J M, Lehman L M, Gudiol F, Gonzales J, Raventos A, Rubio R, Bouza E, Pintado V, Gil Aguado A, Garci de Lomas J, Delgado R, Borleffs J C C, Hsu A, Valdes J M, Boucher C A B, Cooper D A. A short term study of the safety, pharmacokinetics and efficacy of ritonavir, an inhibitor of HIV-1 protease. N Engl J Med. 1995;333:1528–1533. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

