Conditional lineage ablation to model human diseases
- PMID: 9736743
- PMCID: PMC21649
- DOI: 10.1073/pnas.95.19.11371
Conditional lineage ablation to model human diseases
Abstract
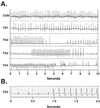
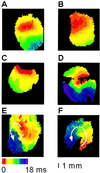
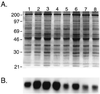
Cell loss contributes to the pathogenesis of many inherited and acquired human diseases. We have developed a system to conditionally ablate cells of any lineage and developmental stage in the mouse by regulated expression of the diphtheria toxin A (DTA) gene by using tetracycline-responsive promoters. As an example of this approach, we targeted expression of DTA to the hearts of adult mice to model structural abnormalities commonly observed in human cardiomyopathies. Induction of DTA expression resulted in cell loss, fibrosis, and chamber dilatation. As in many human cardiomyopathies, transgenic mice developed spontaneous arrhythmias in vivo, and programmed electrical stimulation of isolated-perfused transgenic hearts demonstrated a strikingly high incidence of spontaneous and inducible ventricular tachycardia. Affected mice showed marked perturbations of cardiac gap junction channel expression and localization, including a subset with disorganized epicardial activation patterns as revealed by optical action potential mapping. These studies provide important insights into mechanisms of arrhythmogenesis and suggest that conditional lineage ablation may have wide applicability for studies of disease pathogenesis.
Figures
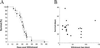



References
-
- Hetts S W. J Am Med Assoc. 1998;279:300–307. - PubMed
-
- Rinkenberger J L, Korsmeyer S J. Curr Opin Genet Dev. 1997;7:589–596. - PubMed
-
- MacLellan W R, Schneider M D. Circ Res. 1997;81:137–144. - PubMed
-
- Barbosa E R, Limongi J C, Cummings J L. Psychiatr Clin North Am. 1997;20:769–790. - PubMed
-
- Lev M. Cardiovasc Clin. 1972;4:159–186. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

