Activation of the genetically defined m1 muscarinic receptor potentiates N-methyl-D-aspartate (NMDA) receptor currents in hippocampal pyramidal cells
- PMID: 9736760
- PMCID: PMC21666
- DOI: 10.1073/pnas.95.19.11465
Activation of the genetically defined m1 muscarinic receptor potentiates N-methyl-D-aspartate (NMDA) receptor currents in hippocampal pyramidal cells
Abstract
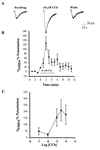
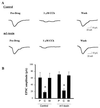
Evidence suggests that cholinergic input to the hippocampus plays an important role in learning and memory and that degeneration of cholinergic terminals in the hippocampus may contribute to the memory loss associated with Alzheimer's disease. One of the more prominent effects of cholinergic agonists on hippocampal physiology is the potentiation of N-methyl-D-aspartate (NMDA)-receptor currents by muscarinic agonists. Here, we employ traditional pharmacological reagents as well as m1-toxin, an m1 antagonist with unprecedented selectivity, to demonstrate that this potentiation of NMDA-receptor currents in hippocampal CA1 pyramidal cells is mediated by the genetically defined m1 muscarinic receptor. Furthermore, we demonstrate the colocalization of the m1 muscarinic receptor and the NR1a NMDA receptor subunit at the electron microscopic level, indicating a spatial relationship that would allow for physiological interactions between these two receptors. This work demonstrates that the m1-muscarinic receptor gene product modulates excitatory synaptic transmission, and it has important implications in the study of learning and memory as well as the design of drugs to treat neurodegenerative diseases such as Alzheimer's.
Figures



References
-
- Brown T H, Zador A M. In: The Synaptic Organization of the Brain. Shepherd G M, editor. New York: Oxford Univ. Press; 1990. pp. 346–388.
-
- Drachman D A, Leavitt J L. Arch Neurol. 1974;30:113–121. - PubMed
-
- Bartus R T, Dean D L I, Beer B, Lippa A S. Science. 1982;217:783–790. - PubMed
-
- Dekker A J A M, Connor D J, Thal L J. Neurosci Biobehav Rev. 1991;15:299–317. - PubMed
-
- Fibiger H C. Trends Neurosci. 1991;14:220–223. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

