Ultrasensitive reverse transcription-PCR assay for quantitation of human immunodeficiency virus type 1 RNA in plasma
- PMID: 9738051
- PMCID: PMC105095
- DOI: 10.1128/JCM.36.10.2964-2969.1998
Ultrasensitive reverse transcription-PCR assay for quantitation of human immunodeficiency virus type 1 RNA in plasma
Abstract
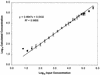
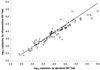
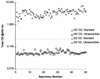
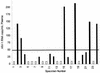
With the recent introduction of combination therapy, human immunodeficiency virus type 1 (HIV-1) RNA levels in plasma have been dramatically reduced, frequently to below the limit of quantitation (400 copies/ml of plasma) of the AMPLICOR HIV-1 MONITOR Test (Roche Diagnostic Systems). To achieve enhanced sensitivity of the AMPLICOR HIV-1 MONITOR Test, a modified specimen preparation procedure that allows input of RNA from 10-fold more plasma per amplification reaction was developed. This "ultrasensitive" method allows the accurate quantitation of plasma HIV-1 RNA levels as low as 50 copies/ml. A precision study yielded average within-run and between-run coefficients of variation (CV) of 24.8 and 9.6%, respectively. A multicenter reproducibility study demonstrated that the laboratory-to-laboratory reproducibility of this assay is good, with an average CV of 32%. The linear range of this test is between 50 and 50,000 copies/ml of plasma. RNA concentrations measured by the ultrasensitive and standard HIV-1 MONITOR tests exhibited good agreement within the shared linear range of the two methods. The two measurements were within a factor of 2 for 91% of the specimens tested, with the concentration measured by the ultrasensitive method being only slightly lower (median, 22% lower). Preliminary studies suggest that this assay will prove to be useful for predicting the stability of viral suppression in patients whose RNA levels drop below 400 copies/ml in response to highly active antiretroviral therapy.
Figures
Similar articles
-
Quantitative and cost comparison of ultrasensitive human immunodeficiency virus type 1 RNA viral load assays: Bayer bDNA quantiplex versions 3.0 and 2.0 and Roche PCR Amplicor monitor version 1.5.J Clin Microbiol. 2000 Mar;38(3):1113-20. doi: 10.1128/JCM.38.3.1113-1120.2000. J Clin Microbiol. 2000. PMID: 10699005 Free PMC article.
-
Performance characteristics of the QUANTIPLEX HIV-1 RNA 3.0 assay for detection and quantitation of human immunodeficiency virus type 1 RNA in plasma.J Clin Microbiol. 2000 Aug;38(8):2837-45. doi: 10.1128/JCM.38.8.2837-2845.2000. J Clin Microbiol. 2000. PMID: 10921936 Free PMC article.
-
A new ultrasensitive assay for quantitation of HIV-1 RNA in plasma.J Virol Methods. 2001 Jun;95(1-2):81-92. doi: 10.1016/s0166-0934(01)00297-x. J Virol Methods. 2001. PMID: 11377715
-
Comparative evaluation of the Cobas Amplicor HIV-1 Monitor Ultrasensitive Test, the new Cobas AmpliPrep/Cobas Amplicor HIV-1 Monitor Ultrasensitive Test and the Versant HIV RNA 3.0 assays for quantitation of HIV-1 RNA in plasma samples.J Clin Virol. 2005 May;33(1):43-51. doi: 10.1016/j.jcv.2004.09.025. J Clin Virol. 2005. PMID: 15797364
-
Evaluation of the ultrasensitive Roche Amplicor HIV-1 monitor assay for quantitation of human immunodeficiency virus type 1 RNA.J Clin Microbiol. 1999 Mar;37(3):792-5. doi: 10.1128/JCM.37.3.792-795.1999. J Clin Microbiol. 1999. PMID: 9986856 Free PMC article.
Cited by
-
Quantitation of human immunodeficiency virus type 1 (HIV-1) RNA in cell-free cervicovaginal secretions: comparison of reverse transcription-PCR amplification (AMPLICOR HIV-A MONITOR 1.5) with enhanced-sensitivity branched-DNA assay (Quantiplex 3.0).J Clin Microbiol. 2001 Jun;39(6):2055-9. doi: 10.1128/JCM.39.6.2055-2059.2001. J Clin Microbiol. 2001. PMID: 11376034 Free PMC article.
-
New real-time reverse transcriptase-initiated PCR assay with single-copy sensitivity for human immunodeficiency virus type 1 RNA in plasma.J Clin Microbiol. 2003 Oct;41(10):4531-6. doi: 10.1128/JCM.41.10.4531-4536.2003. J Clin Microbiol. 2003. PMID: 14532178 Free PMC article.
-
Comparison of versions 1.0 AND 1.5 of the UltraSensitive AMPLICOR HIV-1 MONITOR test for subjects with low viral load.J Clin Microbiol. 2004 Jun;42(6):2774-6. doi: 10.1128/JCM.42.6.2774-2776.2004. J Clin Microbiol. 2004. PMID: 15184467 Free PMC article.
-
HIV-1 genotypic resistance patterns predict response to saquinavir-ritonavir therapy in patients in whom previous protease inhibitor therapy had failed.Ann Intern Med. 1999 Dec 7;131(11):813-21. doi: 10.7326/0003-4819-131-11-199912070-00003. Ann Intern Med. 1999. PMID: 10610625 Free PMC article.
-
A multicenter study evaluation of the digene hybrid capture II signal amplification technique for detection of hepatitis B virus DNA in serum samples and testing of EUROHEP standards.J Clin Microbiol. 2000 Jun;38(6):2150-5. doi: 10.1128/JCM.38.6.2150-2155.2000. J Clin Microbiol. 2000. PMID: 10834968 Free PMC article. Clinical Trial.
References
-
- BHIVA Guidelines Co-ordinating Committee. British HIV Association guidelines for antiretroviral treatment of HIV seropositive individuals. Lancet. 1997;349:1086–1092. - PubMed
-
- Carpenter C C, Fischl M A, Hammer S M, Hirsch M S, Jacobsen D M, Katzenstein D A, Montaner J S, Richman D D, Saag M S, Schooley R T, Thompson M A, Vella S, Yeni P G, Volberding P A for the International AIDS Society-USA. Antiretroviral therapy for HIV infection in 1996. Recommendations of an international panel. JAMA. 1996;276:146–154. - PubMed
-
- Centers for Disease Control and Prevention. Public Health Service task force recommendations for the use of antiretroviral drugs in pregnant women infected with HIV-1 for maternal health and for reducing perinatal HIV-1 transmission in the United States. Morbid Mortal Weekly Rep. 1998;47(No. RR-2):1–39. - PubMed
-
- Centers for Disease Control and Prevention. Guidelines for the use of antiretroviral agents in pediatric HIV infection. Morbid Mortal Weekly Rep. 1998;47(No. RR-4):1–51. - PubMed
-
- Eron J J, Benoit S L, Jemsek J, MacArthur R D, Santana J, Quinn J B, Kuritzkes D R, Fallon M A, Rubin and the North American HIV Working Party M. Treatment with lamivudine, zidovudine, or both in HIV-positive patients with 200 to 500 CD4+ cells per cubic millimeter. N Engl J Med. 1995;333:1662–1669. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

