Brain-derived neurotrophic factor and basic fibroblast growth factor downregulate NMDA receptor function in cerebellar granule cells
- PMID: 9742162
- PMCID: PMC6792992
- DOI: 10.1523/JNEUROSCI.18-19-07953.1998
Brain-derived neurotrophic factor and basic fibroblast growth factor downregulate NMDA receptor function in cerebellar granule cells
Abstract
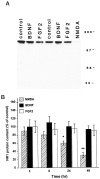
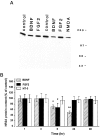
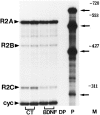
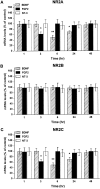
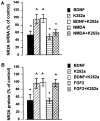
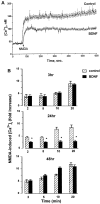
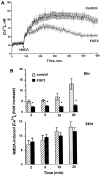
Evidence has accumulated to suggest that the NMDA glutamate receptor subtype plays an important role in neuronal degeneration evoked by hypoxia, ischemia, or trauma. Cerebellar granule cells in culture are vulnerable to NMDA-induced neuronal excitotoxicity. In these cells, brain-derived neurotrophic factor (BDNF) and basic fibroblast growth factor (FGF2) prevent the excitotoxic effect of NMDA. However, little is known about the molecular mechanisms underlying the protective properties of these trophic factors. Using cultured rat cerebellar granule cells, we investigated whether BDNF and FGF2 prevent NMDA toxicity by downregulating NMDA receptor function. Western blot and RNase protection analyses were used to determine the expression of the various NMDA receptor subunits (NR1, NR2A, NR2B, and NR2C) after BDNF or FGF2 treatment. FGF2 and BDNF elicited a time-dependent decrease in the expression of NR2A and NR2C subunits. Because NMDA receptor activation leads to increased intracellular Ca2+ concentration ([Ca2+]i), we studied the effect of the BDNF- and FGF2-induced reduction in NR2A and NR2C synthesis on the NMDA-evoked Ca2+ responses by single-cell fura-2 fluorescence ratio imaging. BDNF and FGF2 reduced the NMDA-mediated [Ca2+]i increase with a time dependency that correlates with their ability to decrease NR2A and NR2C subunit expression, suggesting that these trophic factors also induce a functional downregulation of the NMDA receptor. Because sustained [Ca2+]i is believed to be causally related to neuronal injury, we suggest that BDNF and FGF2 may protect cerebellar granule cells against excitotoxicity by altering the NMDA receptor-Ca2+ signaling via a downregulation of NMDA receptor subunit expression.
Figures
References
-
- Anegawa NJ, Lynch DR, Verdoorn TA, Pritchett DB. Transfection of N-methyl-d-aspartate receptors in a nonneuronal cell line leads to cell death. J Neurochem. 1995;64:2004–2012. - PubMed
-
- Berg MM, Sterneberg DW, Parada LF, Chao MV. K-252a inhibits nerve growth factor-induced trk proto-oncogene tyrosine phosphorylation and kinase activity. J Biol Chem. 1992;267:13–16. - PubMed
-
- Burgoyne RD, Cambray-Deakin MA. The cellular neurobiology of neuronal development: the cerebellar granule cell. Brain Res Rev. 1988;13:77–101. - PubMed
-
- Chao M, Hempstead B. p75 and Trk: a two-receptor system. Trends Neurosci. 1995;18:321–326. - PubMed
-
- Cheng B, Furukawa K, O’Keefe JA, Goodman Y, Kihiko M, Fabian T, Mattson MP. Basic fibroblast growth factor selectively increases AMPA-receptor subunit GluR1 protein level and differentially modulates Ca2+ responses to AMPA and NMDA in hippocampal neurons. J Neurochem. 1995;65:2525–2536. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
