Inhibition of angiogenesis by interleukin 4
- PMID: 9743522
- PMCID: PMC2212547
- DOI: 10.1084/jem.188.6.1039
Inhibition of angiogenesis by interleukin 4
Abstract
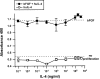
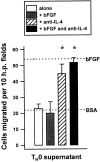
Interleukin (IL)-4, a crucial modulator of the immune system and an active antitumor agent, is also a potent inhibitor of angiogenesis. When incorporated at concentrations of 10 ng/ml or more into pellets implanted into the rat cornea or when delivered systemically to the mouse by intraperitoneal injection, IL-4 blocked the induction of corneal neovascularization by basic fibroblast growth factor. IL-4 as well as IL-13 inhibited the migration of cultured bovine or human microvascular cells, showing unusual dose-response curves that were sharply stimulatory at a concentration of 0.01 ng/ml but inhibitory over a wide range of higher concentrations. Recombinant cytokine from mouse and from human worked equally well in vitro on bovine and human endothelial cells and in vivo in the rat, showing no species specificity. IL-4 was secreted at inhibitory levels by activated murine T helper (TH0) cells and by a line of carcinoma cells whose tumorigenicity is known to be inhibited by IL-4. Its ability to cause media conditioned by these cells to be antiangiogenic suggested that the antiangiogenic activity of IL-4 may play a role in normal physiology and contribute significantly to its demonstrated antitumor activity.
Figures







References
-
- Paul WE. Interleukin-4: a prototypic immunoregulatory lymphokine. Blood. 1991;77:1859–1870. - PubMed
-
- Kopf M, Le Gros G, Bachmann M, Lamers KC, Bluethmann H, Kohler G. Disruption of the murine IL-4 gene blocks Th2 cytokine responses. Nature. 1993;362:245–248. - PubMed
-
- Kuhn R, Rajewsky K, Muller W. Generation and analysis of interleukin-4 deficient mice. Science. 1991;254:707–710. - PubMed
-
- Ohara J, Paul WE. Receptors for B-cell stimulatory factor-1 expressed on cells of haematopoietic lineage. Nature. 1987;325:537–540. - PubMed
-
- Mosmann TR, Yokota T, Kastelein R, Zurawski SM, Arai N, Takebe Y. Species-specificity of T cell stimulating activities of IL 2 and BSF-1 (IL-4): comparison of normal and recombinant, mouse and human IL-2 and BSF-1 (IL-4) J Immunology. 1987;138:1813–1816. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

