The plant cDNA LCT1 mediates the uptake of calcium and cadmium in yeast
- PMID: 9751787
- PMCID: PMC21762
- DOI: 10.1073/pnas.95.20.12043
The plant cDNA LCT1 mediates the uptake of calcium and cadmium in yeast
Abstract
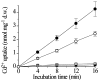
Nonessential metal ions such as cadmium are most likely transported across plant membranes via transporters for essential cations. To identify possible pathways for Cd2+ transport we tested putative plant cation transporters for Cd2+ uptake activity by expressing cDNAs in Saccharomyces cerevisiae and found that expression of one clone, LCT1, renders the growth of yeast more sensitive to cadmium. Ion flux assays showed that Cd2+ sensitivity is correlated with an increase in Cd2+ uptake. LCT1-dependent Cd2+ uptake is saturable, lies in the high-affinity range (apparent KM for Cd2+ = 33 microM) and is sensitive to block by La3+ and Ca2+. Growth assays demonstrated a sensitivity of LCT1-expressing yeast cells to extracellular millimolar Ca2+ concentrations. LCT1-dependent increase in Ca2+ uptake correlated with the observed phenotype. Furthermore, LCT1 complements a yeast disruption mutant in the MID1 gene, a non-LCT1-homologous yeast gene encoding a membrane Ca2+ influx system required for recovery from the mating response. We conclude that LCT1 mediates the uptake of Ca2+ and Cd2+ in yeast and may therefore represent a first plant cDNA encoding a plant Ca2+ uptake or an organellar Ca2+ transport pathway in plants and may contribute to transport of the toxic metal Cd2+ across plant membranes.
Figures







References
-
- Bush D S. Annu Rev Plant Physiol Plant Mol Biol. 1995;46:95–122.
-
- Webb A A R, McAinsh M R, Taylor J E, Hetherington A M. Adv Bot Res. 1996;22:45–96.
-
- Sentenac H, Bonneaud N, Minet M, Lacroute F, Salmon J, Gaymard F, Grignon C. Science. 1992;256:663–665. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

