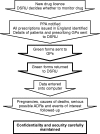
Prescription-event monitoring--recent progress and future horizons
- PMID: 9764958
- PMCID: PMC1873686
- DOI: 10.1046/j.1365-2125.1998.00774.x
Prescription-event monitoring--recent progress and future horizons
Figures
References
-
- Committee on Safety of Drugs. Report for 1969 and 1970. London: HMSO; 1971.
-
- Mann RD. The yellow card data: the nature and scale of the adverse drug reactions problems. In: Mann RD, editor. Adverse drug reactions. Carnforth, UK: Parthenon Publishing; 1987. pp. 62–63.
-
- Freemantle SN, Pearce GL, Wilton LV, Mackay FJ, Mann RD. The incidence of the most commonly reported events with 40newly marketed drugs—a study by Prescription-Event Monitoring. Pharmacoepidemiology and Drug Safety. 1997;6:1.
-
- British Medical Association. Reporting Adverse Drug Reactions. A BMA policy document. London: British Medical Association; 1996.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources