The contribution of pH-dependent mechanisms to fatigue at different intensities in mammalian single muscle fibres
- PMID: 9769425
- PMCID: PMC2231252
- DOI: 10.1111/j.1469-7793.1998.831bd.x
The contribution of pH-dependent mechanisms to fatigue at different intensities in mammalian single muscle fibres
Abstract
1. The contribution of intracellular pH (pHi) to the failure of Ca2+ release and inhibition of contractile proteins observed during fatigue was assessed in single intact mouse muscle fibres at 22 C. Fatigue was induced by repeated tetani at intensities designed to induce different levels of intracellular acidosis. Force and either intracellular free Ca2+ concentration ([Ca2+]i; measured using indo-1) or pHi (measured using SNARF-1) were recorded in fibres fatigued at two different intensities. 2. Intensity was varied by the repetition rate of tetani and quantified by the duty cycle (the fraction of time when the muscle was tetanized). Stimulation at the low intensity (duty cycle approximately 0.1) reduced force to 30 % of initial values in 206 +/- 21 s (60 +/- 7 tetani); at the high intensity (duty cycle approximately 0.3) force was reduced to 30% in 42 +/- 7 s (43 +/- 7 tetani) (P < 0.05; n = 14). 3. When force was reduced to 30 % of initial values, tetanic [Ca2+]i had fallen from 648 +/- 87 to 336 +/- 64 nM (48% decrease) at the low intensity but had only fallen from 722 +/- 84 to 468 +/- 60 nM (35% decrease) at the higher intensity (P < 0.05 low vs. high intensity; n = 7). 4. Fatigue resulted in reductions in Ca2+ sensitivity of the contractile proteins which were greater at the high intensity (pre-fatigue [Ca2+]i required for 50 % of maximum force (Ca50) = 354 +/- 23 nM; post-fatigue Ca50 = 421 +/- 48 nM and 524 +/- 43 nM for low and high intensities, respectively). Reductions in maximum Ca2+-activated force (Fmax) were similar at the two intensities (pre-fatigue Fmax = 328 +/- 22 microN; post-fatigue Fmax = 271 +/- 20 and 265 +/- 19 microN for low and high intensities, respectively). 5. Resting pHi was 7.15 +/- 0.05. During fatigue at the low intensity, pHi was reduced by 0.12 +/- 0.02 pH units and at the high intensity pHi was reduced by 0.34 +/- 0.07 pH units (P < 0.05; n = 5). 6. Our results indicate that the more rapid fall in force at a high intensity is due to a reduction in Ca2+ sensitivity of the contractile proteins, probably related to the greater acidosis. Our data also indicate that the failure of Ca2+ release and reduced maximum Ca2+-activated force observed during fatigue are not due to reductions in intracellular pH.
Figures
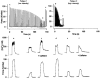

 ), minimum force (
), minimum force ( ) and after 5 mm caffeine exposure at the end of fatigue (
) and after 5 mm caffeine exposure at the end of fatigue ( ). Minimum force was defined as the force immediately before a response to caffeine was observed. Changes in force and [Ca2+]i during fatigue at both the low intensity (1/4 s, etc.) and high intensity (1/1 s) are shown. Values represent mean ± s.e.m. (n = 7). *P < 0.05 vs. pre-fatigue; †P < 0.05 vs. fatigue; ‡P < 0.05 vs. low intensity.
). Minimum force was defined as the force immediately before a response to caffeine was observed. Changes in force and [Ca2+]i during fatigue at both the low intensity (1/4 s, etc.) and high intensity (1/1 s) are shown. Values represent mean ± s.e.m. (n = 7). *P < 0.05 vs. pre-fatigue; †P < 0.05 vs. fatigue; ‡P < 0.05 vs. low intensity.
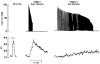

 and
and  , respectively). Reductions in force and [Ca2+]i, shown as % of initial values, were of similar magnitude at both intensities. Values represent mean ± s.e.m. (n = 6–7). * Significant difference (P < 0.05) compared with initial force or [Ca2+]i at the same frequency or condition.
, respectively). Reductions in force and [Ca2+]i, shown as % of initial values, were of similar magnitude at both intensities. Values represent mean ± s.e.m. (n = 6–7). * Significant difference (P < 0.05) compared with initial force or [Ca2+]i at the same frequency or condition.References
-
- Allen DG, Duty S, Westerblad H. Metabolic changes in muscle during exercise: their effects on muscle function. Proceedings of the Australian Physiological and Pharmacological Society. 1993;24:65–75.
-
- Allen DG, Lännergrenauml;nnergren J, Westerblad H. Muscle cell function during prolonged activity: cellular mechanisms of fatigue. Experimental Physiology. 1995a;80:497–527. - PubMed
-
- Allen DG, Westerblad H, Lännergrenauml;nnergren J. The role of intracellular acidosis in muscle fatigue. Advances in Experimental Medicine and Biology. 1995b;384:57–68. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

