Development of a new cytomegalovirus (CMV) immunoglobulin M (IgM) immunoblot for detection of CMV-specific IgM
- PMID: 9774589
- PMCID: PMC105325
- DOI: 10.1128/JCM.36.11.3337-3341.1998
Development of a new cytomegalovirus (CMV) immunoglobulin M (IgM) immunoblot for detection of CMV-specific IgM
Abstract
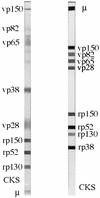
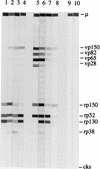
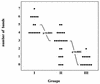
We developed a new cytomegalovirus (CMV) immunoglobulin M (IgM) immunoblot to detect CMV-specific IgM in human sera. The new test contains four viral proteins (vp150, vp82, vp65, and vp28) purified from viral particles and four recombinant proteins (rp150, rp130, rp52, and rp38) purified from Escherichia coli. These antigens were individually loaded onto nitrocellulose strips, and the strips were then used to detect CMV-specific IgM by using a mu-specific conjugate. The new assay was evaluated in parallel with one or two IgM enzyme-linked immunosorbent assays (ELISAs) to test 592 serum samples from different groups of latently or acutely infected individuals. The sensitivity of the new assay with respect to the consensus of two ELISAs was 100%, the specificity was 98.6%, the positive predictive value was 96.9%, and the negative predictive value was 100%. We also evaluated the new test by testing sera from pregnant women and transplant recipients with a known clinical history. Our results suggest that the new test combines high sensitivity with high specificity, characteristics that are mutually exclusive with the other commercially available tests. Furthermore, a statistically significant correlation was observed between the number of IgM-reactive bands and the elevated risk of transmission from CMV-infected pregnant women to their offspring.
Figures
References
-
- Basson J, Tardy J C, Aymard M. Pattern of anti-cytomegalovirus IgM antibodies determined by immunoblotting. A study of kidney graft recipients developing a primary or recurrent CMV infection. Arch Virol. 1989;108:259–270. - PubMed
-
- Boppana S B, Britt W J. Antiviral antibody responses and intrauterine transmission after primary maternal cytomegalovirus infection. J Infect Dis. 1995;171:1115–1121. - PubMed
-
- Braun W, Weber B, Moell U, Hamann A, Doerr H W. Immunoglobulin A and M patterns to human cytomegalovirus during recurrent infection in patients with AIDS using a modified Western blot. J Virol Methods. 1993;43:65–76. - PubMed
-
- Chee M S, Bankier A T, Beck S, Bohni R, Brown C M, Cerny T, Horsnell T, Hutchinson C A, Kouzarides T, Martinetti J A. Analysis of the protein-coding content of the sequence of human cytomegalovirus strain AD 169. Curr Top Microbiol Immunol. 1990;154:125–169. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

